CHEM 112 Lecture Notes - Lecture 2: Nitrous Acid, Chemical Equilibrium, Buffer Solution
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
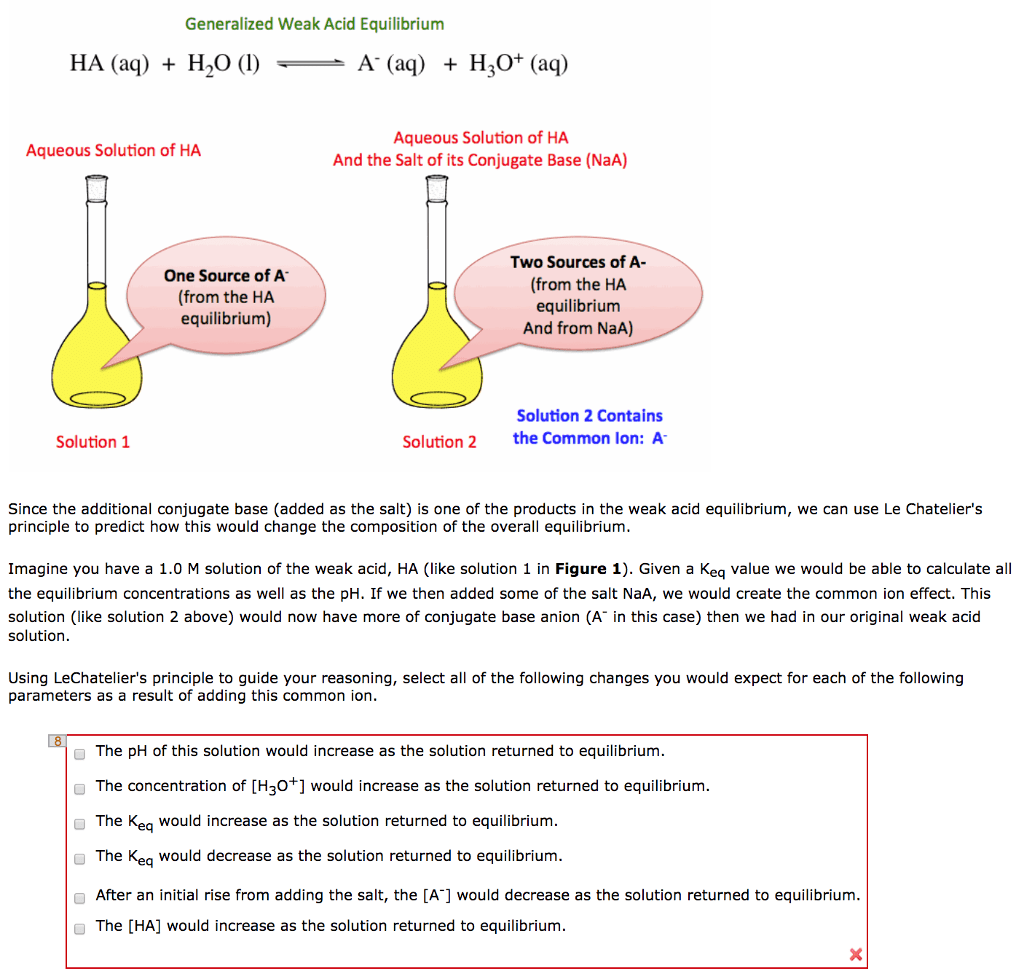
Common Ion Effect
A) Mg(OH)2 is a sparingly soluble compound, in this case a base, with a solubility product, Ksp, of 5.61Ã10â11. It is used to control the pH and provide nutrients in the biological (microbial) treatment of municipal wastewater streams.
Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in pure H2O?
Express your answer with the appropriate units.
View Available Hint(s)
| | |||
| molar solubility = |
Submit
The common-ion effect and solubility
The solubility of a slightly soluble compound can be greatly affected by the addition of a soluble compound with a common ion, that is, with one of the ions in the added soluble compound being identical to one of the ions of the slightly soluble compound. The general result of the addition of the common ion is to greatly reduce the solubility of the slightly soluble compound. In other words, the addition of the common ion results in a shift in the equilibrium of the slightly soluble compound.
Part B - Calculate the molar solubility in NaOH
Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in 0.200 M NaOH?
Express your answer with the appropriate units.
View Available Hint(s)
| | |||
| molar solubility = |
Submit
Part C
This question will be shown after you complete previous question(s).
The common-ion effect and buffer systems
A buffer is a mixed solution of a weak acid or base, combined with its conjugate. Note that this can be understood essentially as a common-ion problem: The conjugate is a common ion added to an equilibrium system of a weak acid or base. The addition of the conjugate shifts the equilibrium of the system to relieve the stress of the added concentration of the common ion. In a solution consisting of a weak acid or base, the equilibrium shift also results in a pH shift of the system.
It is the presence of the common ion in the system that results in buffering behavior, because both added H+ or OHâ ions can be neutralized.
Part D
What is the pH change of a 0.290 M solution of citric acid (pKa=4.77) if citrate is added to a concentration of 0.145 M with no change in volume?
Express the difference in pH numerically to two decimal places.
View Available Hint(s)
| |