CHEM261 Lecture Notes - Lecture 5: Concerted Reaction, Hexamethylphosphoramide, Activation Energy

1
CHEM261 Full Course Notes
Verified Note
1 document
Document Summary
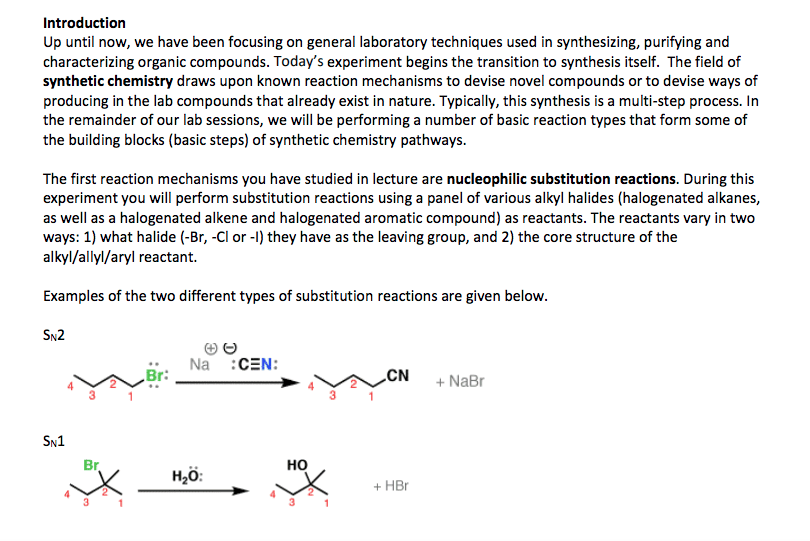
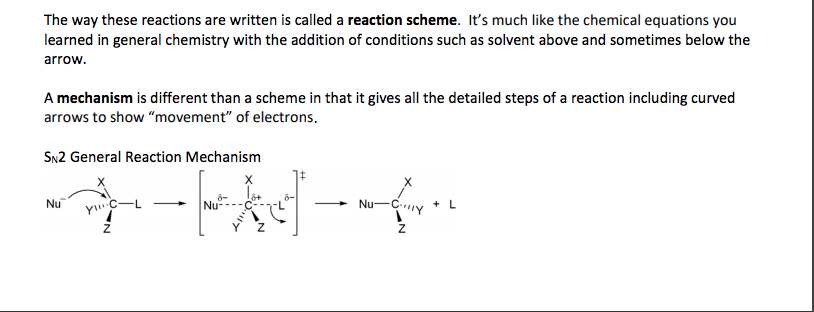
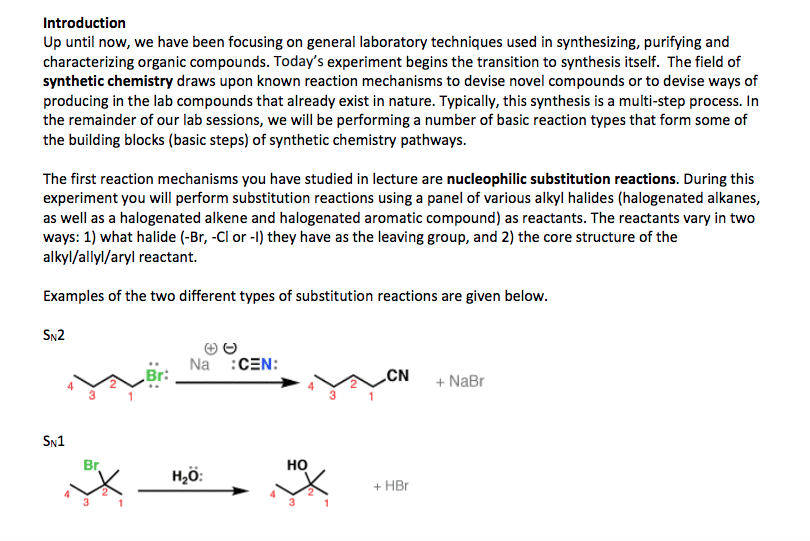
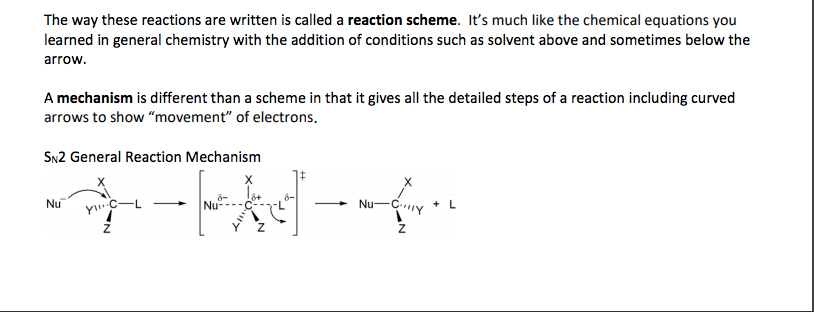
Alkyl halide (haloalkane) general formula is r-x. where x is cl, br or i. Involve substitution and elimination reactions. (two types of elimination and two types of substitution) is en and takes the electrons. Methyl halide = ch3-x. there are primary, secondary and tertiary structures. C-f bonds are too strong for a reaction to occur. Carbon attracts nucleophiles (species with lone pairs of electrons) because the halogen. All reactions lead to elimination of halogen. A nucleophile any negative ion or neutral molecule that has lone-pair of electrons are. Alkyl halide is called the substrate because it is attacked by nucleophile. the halogen. Nucleophilic reaction (sn2 reaction) is attracted to a positive center. it is also a lewis base. (x) is the leaving group. versa) Reactions. same time). backside attack on sp3 carbon. Kinetic studies (order of reactions) are important in the rate determining steps. Reaction is stereospecific (inversion of configuration from r to s and vice.