CHEM123 Lecture Notes - Lecture 3: Stoichiometry, Reaction Quotient, Thermodynamic Equilibrium
108 views6 pages
22 Mar 2016
School
Department
Course
Professor
Document Summary
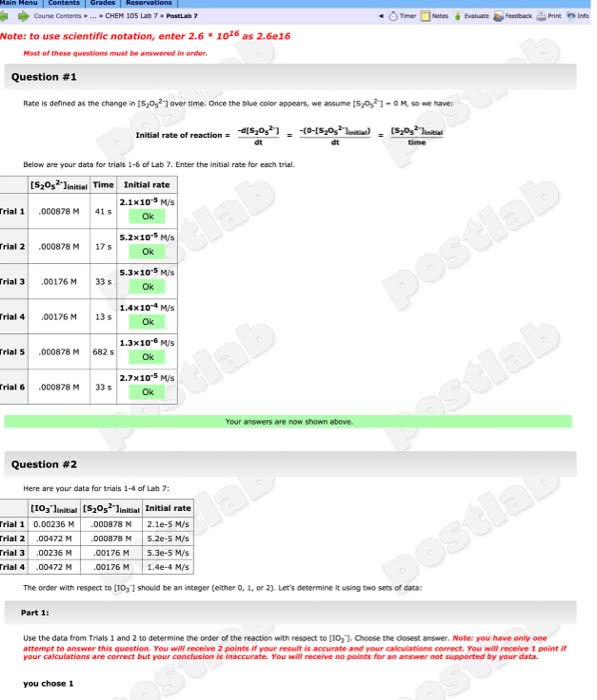
Print off these 9 questions and write out your answers in your own words, as if you were doing an assignment or writing a test which you have to submit. Please note: for the participation quizzes, you are not actually graded on whether your answers are right or wrong, but only on whether or not you participate. The instructor(s) and/or tas review the responses to determine if any conceptual problems they need to address. You have only 1 attempt at the quiz (just like a test or exam). Note: although you are not graded on whether or not your answers to the participation questions are correct, you should make every effort to answer the questions as best as you can. The reaction quotient, qc can be determined from the concentrations of reactant(s) and/or product(s) at any time during a chemical reaction. The values of qc can span: 0 qc .
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232