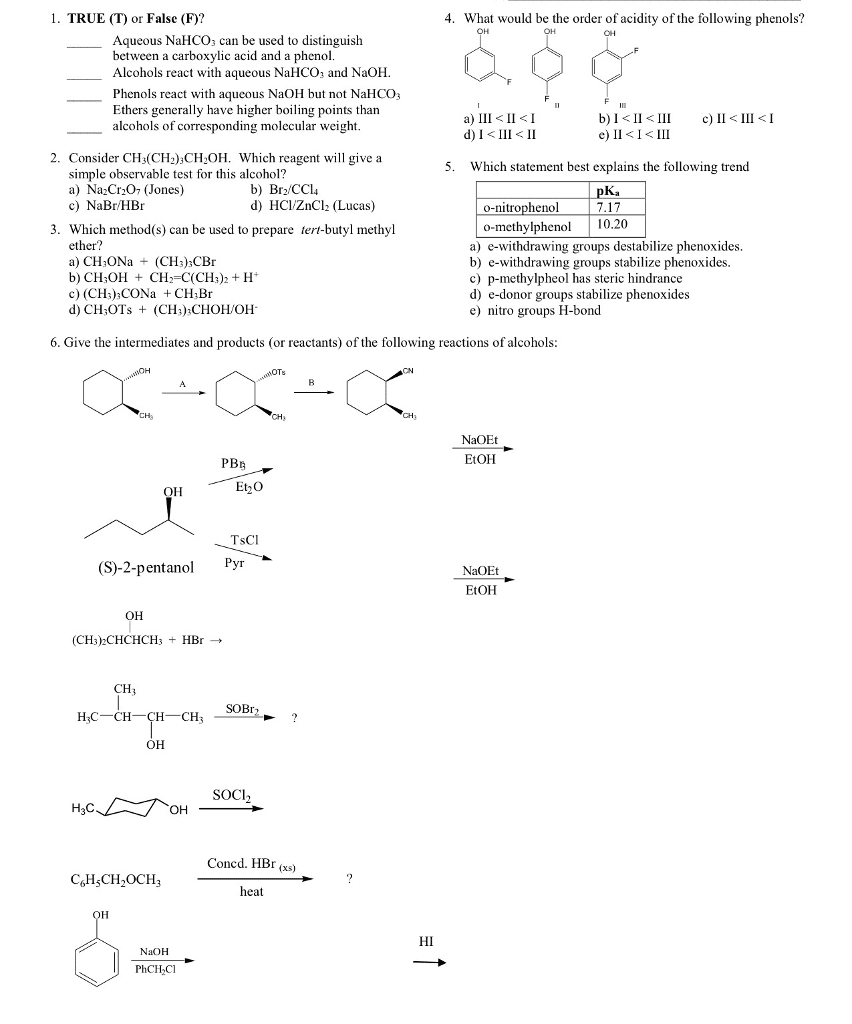
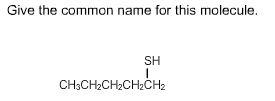
CHM136H1 Lecture Notes - Oxonium Ion, Alkoxide, Phenol

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
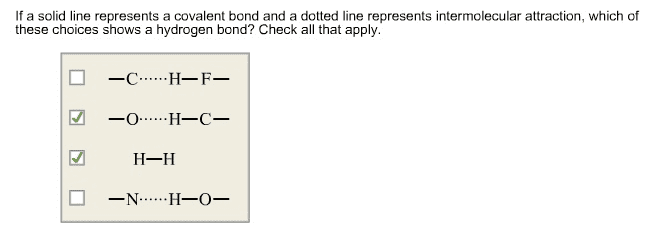
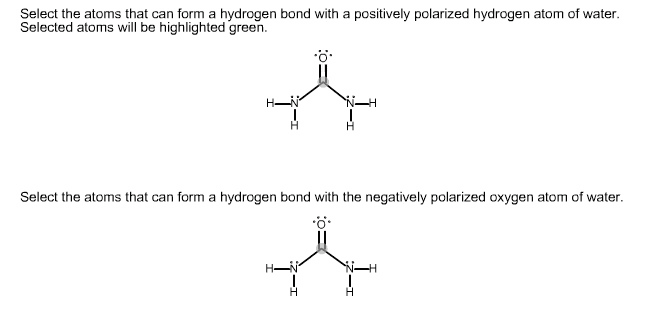
Alcohols and phenols have nearly the same geometry around the oxygen atom as water. R-o-h bond angle has an approximately tetrahedral value and the oxygen is sp3-hybriedized. A positively polarized oh atom is attracted to a lone pair of electron on the en oxygen of another molecule. Intermolecular attraction must be overcome for a molecule to break free from the liquid and enter vapor state. Weakly basic: reversibly protonated by strong acids to yield oxonium ions, roh2+ Weakly acids: dissociate slightly in dilute aqueous solution by donating a proton to water, making h3o+ and an alkoxide ion ro-, or a phenoxide ion, aro- Smaller ka and larger pka are less acidic. Larger ka and smaller pka are more acidic. Inductive effects are also important in determining alcohol acidities. Electron-withdrawing halogen substituents stabilize an alkoxide ion by spreading out the charge over a larger volume, thus making the alcohol more acidic.