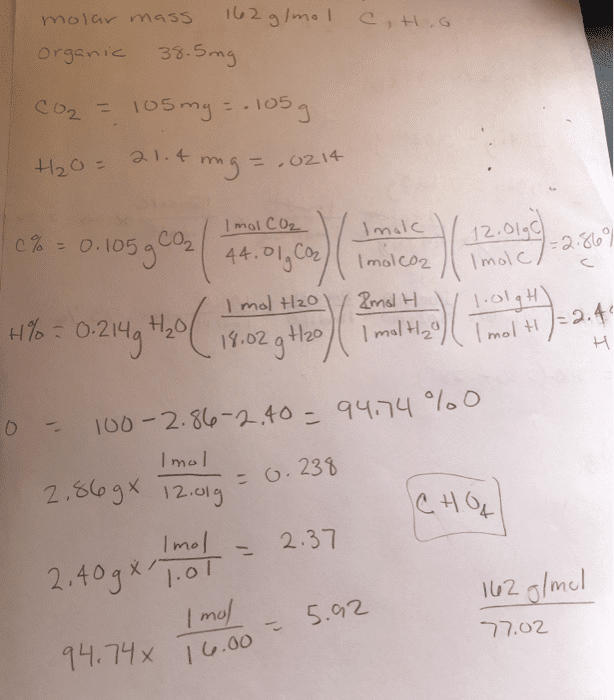Chemistry 1301A/B Lecture Notes - Lecture 30: Mass Spectrometry, Chemical Formula, Molar Mass
Document Summary
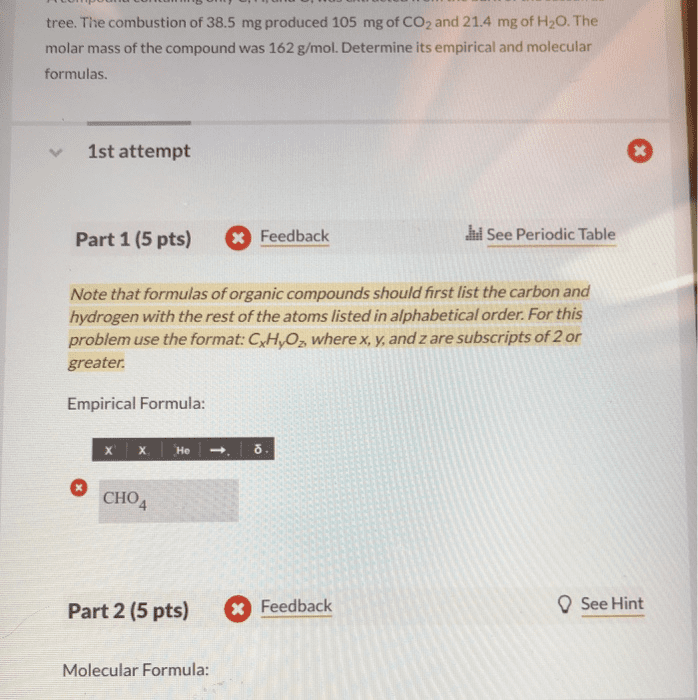
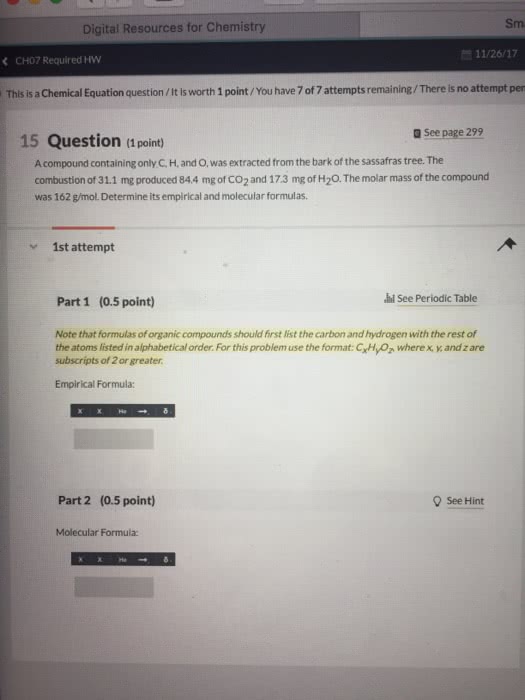
The molecular formula completely and uniquely identifies the unknown compound. This will result in a unit less number: multiply the subscripts of the empirical formula by the unit less number determined in the previous step- the result is the molecular formula. Many compounds consist of carbon, hydrogen, and oxygen. Because of the large number of these types of compounds, and the predictability with which they react, a specific type of analysis is used to determine their formulas. Experimental data, which comes in the form of masses of carbon dioxide and water, is obtained using a carbon-hydrogen combustion analyzer (figure 1). Essentially, the unknown hydrocarbon is reacted with an excess of oxygen, resulting in the predictable products of combustion (carbon dioxide and water). These gaseous products are passed along, with the water absorbed in one holding unit, the carbon dioxide absorbed in the next holding unit, and any excess components absorbed in the final holding unit.