CHE-1102 Lecture Notes - Lecture 13: Rate Equation, Reaction Rate Constant
Get access


Related Documents
Related Questions
For the reaction A+B+CâD+E , the initial reaction rate was measured for various initial concentrations of reactants. The following data were collected:
| Trial | [A] (M ) | [B] (M ) | [C] (M ) | Initial rate (M/s ) |
| 1 | 0.20 | 0.20 | 0.20 | 6.0Ã10â5 |
| 2 | 0.20 | 0.20 | 0.60 | 1.8Ã10â4 |
| 3 | 0.40 | 0.20 | 0.20 | 2.4Ã10â4 |
| 4 | 0.40 | 0.40 | 0.20 | 2.4Ã10â4 |
The rate of a chemical reaction depends on the concentrations of the reactants. For the general reaction between A and B ,
aA+bBâcC+dD
The dependence of the reaction rate on the concentration of each reactant is given by the equation called the rate law:
rate=k[A]^m[B]^n
where k is a proportionality constant called the rate constant. The exponent m determines the reaction order with respect to A , and n determines the reaction order with respect to B . The overall reaction order equals the sum of the exponents (m+n ).
Part A
What is the reaction order with respect to A ?
Express your answer as an integer.
Part B
What is the reaction order with respect to B ?
Express your answer as an integer.
Part C
What is the reaction order with respect to C ?
Express your answer as an integer.
Learning Goal:
To understand reaction order and rate constants.
For the general equation aA+bBâcC+dD, the rate law is expressed as follows:
rate=k[A]m[B]n
where m and n indicate the order of the reaction with respect to each reactant and must be determined experimentally and k is the rate constant, which is specific to each reaction.
Order
For a particular reaction, aA+bB+cCâdD, the rate law was experimentally determined to be
rate=k[A]0[B]1[C]2=k[B][C]2
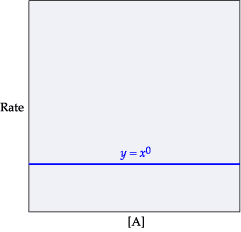
This equation is zero order with respect to A. Therefore, changing the concentration of A has no effect on the rate because [A]0 will always equal 1.(Figure 1)
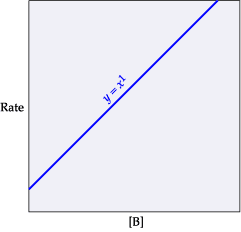
This equation is first order with respect to B. This means that if the concentration of B is doubled, the rate will double. If [B] is reduced by half, the rate will be halved. If [B] is tripled, the rate will triple, and so on.(Figure 2)
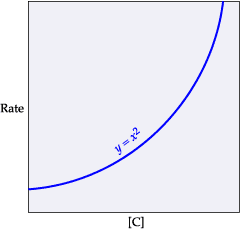
This equation is second order with respect to C. This means that if the concentration of C is doubled, the rate will quadruple. If [C] is tripled, the rate will increase by a factor of 9, and so on.
Overall reaction order and rate-constant units
The sum of the individual orders gives the overall reaction order. The example equation above is third order overall because 0+1+2=3.
For the units of rate to come out to be M/s, the units of the rate constant for third-order reactions must be Mâ2â sâ1 since
M/s=(Mâ2â sâ1)(M3)
For a second-order reaction, the rate constant has units of Mâ1â sâ1 because M/s=(Mâ1â sâ1)(M2). In a first-order reaction, the rate constant has the units sâ1because M/s=(sâ1)(M1).
Analyzing a specific reaction
Consider the following reaction:
2H2+2NOâN2+2H2O,rate=k[H2][NO]2
Part A
What is the overall reaction order?
Express your answer as an integer.
What are the units of the rate constant k for this reaction?
choose one
What are the units of the rate constant for this reaction?
| Mâ sâ1 |
| sâ1 |
| Mâ1â sâ1 |
| Mâ2â sâ1 |
| Mâ3â sâ1 |
Part C
What would happen to the rate if [H2] were doubled?
View Available Hint(s)
The rate would
What would happen to the rate if were doubled?
| stay the same. | ||||
| double. | ||||
| triple. | ||||
| quadruple. What would happen to the rate if [NO] were doubled? View Available Hint(s) The rate would What would happen to the rate if were doubled?
|
Rate
Rate IC]