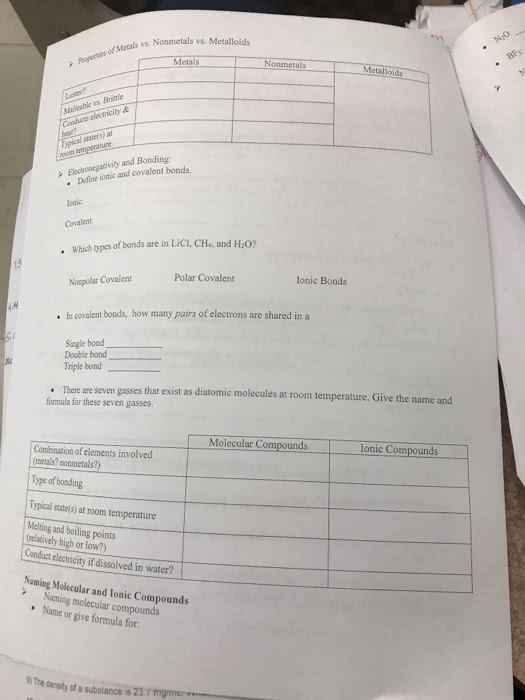
CHM 113 Lecture Notes - Abbreviation, International System Of Units, Kilogram
Document Summary
Chemistry: study of the composition, properties and transformations of matter. Matter- anything that has mass and occupies space. Pure substance matter with fixed composition and distinct properties. Elements: pure substance consisting of just one type of atom, listed by chemical symbol on period table (c for carbon) Compounds: atoms of two or more elements bonded together (h2o) Mixtures two or more pure substances intermingled with one another. Homogeneous mixture (solution): uniform composition throughout, percentage of each component allowed to vary however (air) Physical changes: change in appearance of phase (solid, liquid, gas) but not composition (ice melting, breaking something) Phase changes: freezing (l to s), melting (s to l), vaporization (l to g), condensation (g to l), sublimation (s to g), and deposition (g to s) The temperatures at which the phase changes occur (freezing point, melting point) are called physical properties. Different substances have different sets of physical properties.