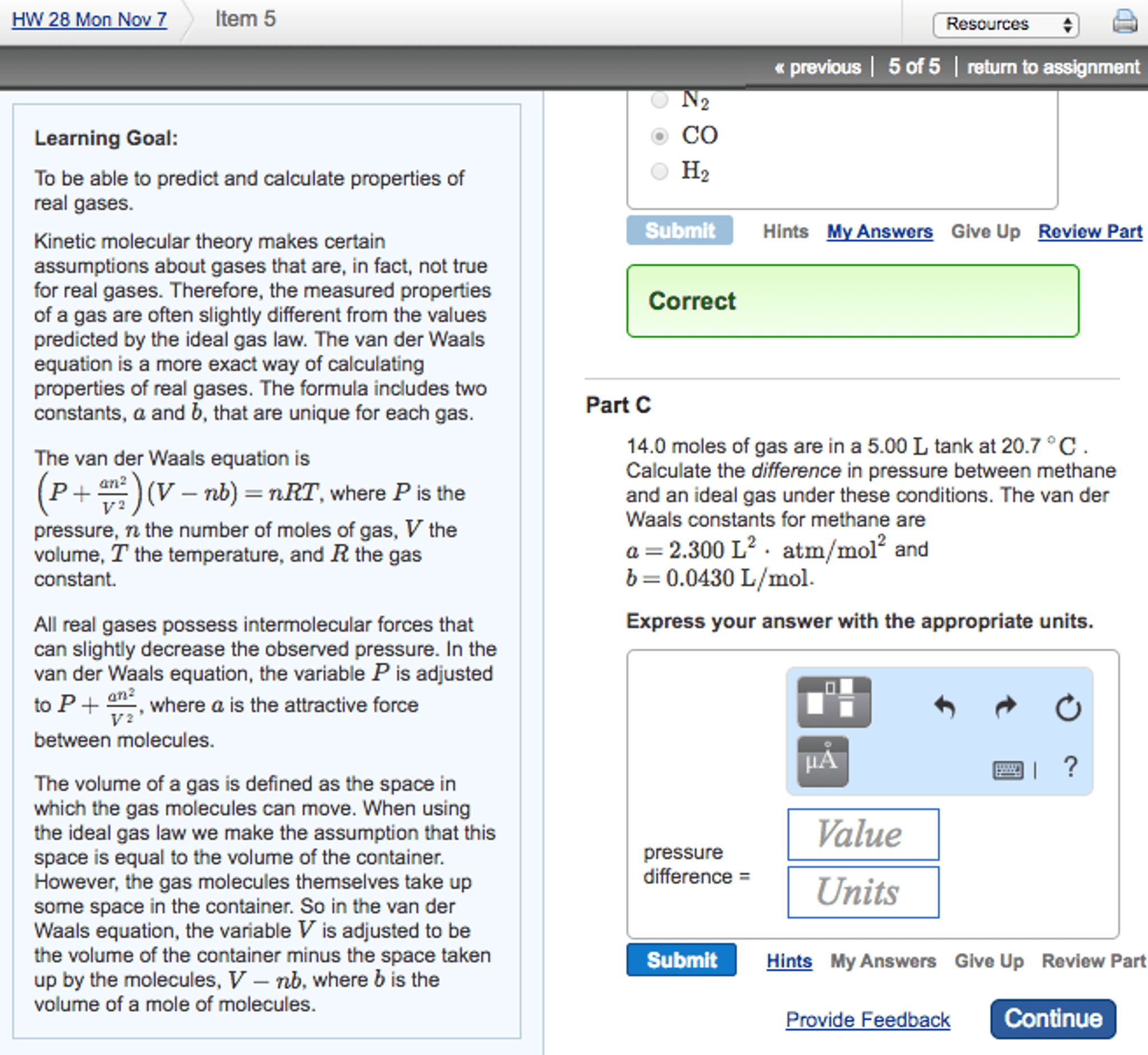
CHM 114 Lecture Notes - Lecture 28: Combined Gas Law, Ideal Gas Law, Hot Air Balloon
Document Summary
Relation between p and v. boyle"s law (1662) V = constant/p or v 1/p (plot volume vs inverse of pressure means linear relationship) Charles" law: v directly proportional to t (fixedn,p) The ratio between the volumes of the reactant gases and the gaseous products can be expressed in simple whole numbers. Provided all volumes are measured at the same t and p. Relation between v and n (avogadro"s hypothesis) Avogadro proposed equal volumes of all gases at the same temp and pressure contain equal number of molecules. Volume directly proportional to number of moles. Boyle"s law, charles" law, avogadro"s law (& amonton"s/gay-lussac"s law) V = 1/p, v = t, v = n, p = t respectively. V = nrt/p (volume of 1 mol of gas at stp) Combined gas law: pv/t = nr = constant. Using the ideal gas law to calculate densities and molar mass. D = m/v = nm/v = pm/rt.