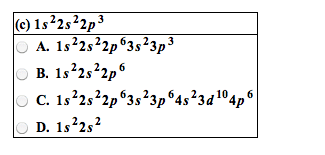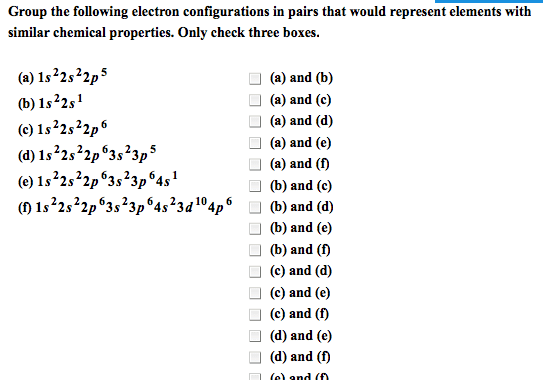CHEM1161 Lecture Notes - Potassium Oxide, Magnesium Nitrate, Sulfate
Document Summary
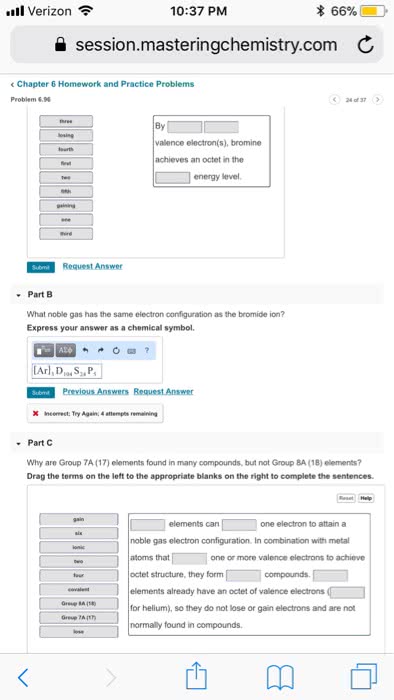
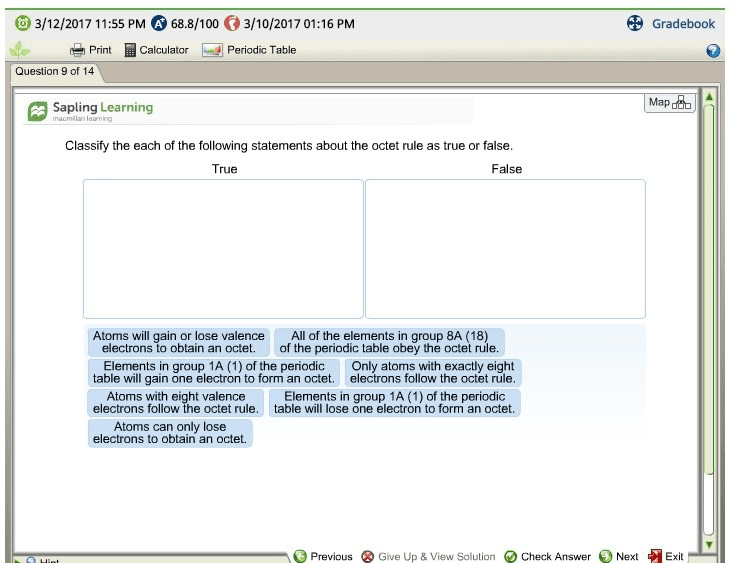
4. 1 octet rule: atoms that are not noble gases form octets, to become more stable, by losing or gaining valence electrons, by sharing valence electrons to form bonds: Metals form positive ions: by losing one or more of their valence electrons, with the electron configuration of the nearest noble gas, with fewer electrons than protons. Group 3a metals ion 3: cation- when an atom loses an electron, metals typically form cations. Nonmetals form negative ions: by gaining one or more valence electrons, with the electron configuration of the nearest noble gas, with fewer protons than electrons. Group 7a nonmetals ion 1 : anions- when an atom gains electrons (nonmetals) Ionic compounds: consist of positive and negative ions, have attractions called ionic bonds between positively and negatively charged ions, have high melting points, are solid at room temperature. In na2s: 2 na atoms lose their valence electrons to become.