CHEM 11200 Lecture Notes - Lecture 1: Bohr Model
24 views1 pages
19 Jul 2020
School
Department
Course
Professor
Document Summary
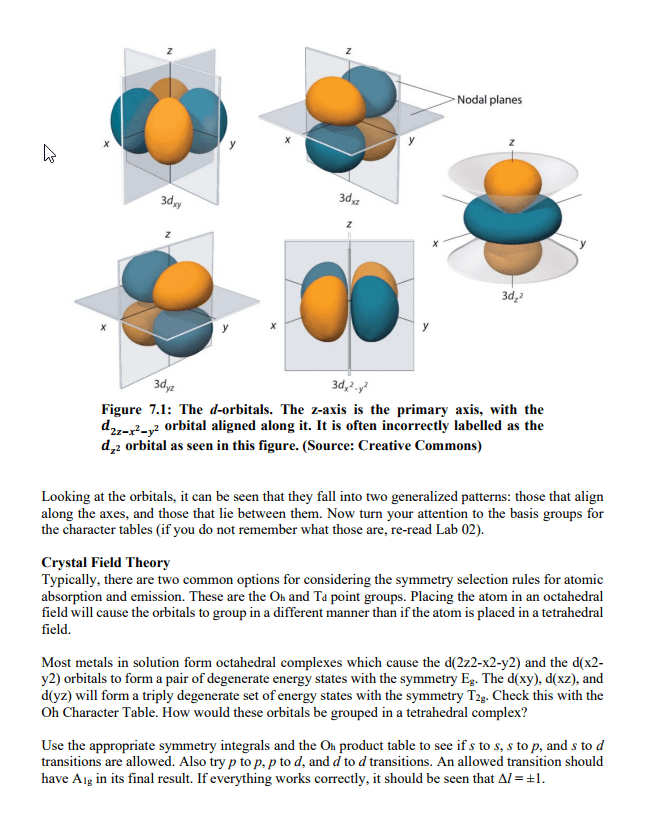
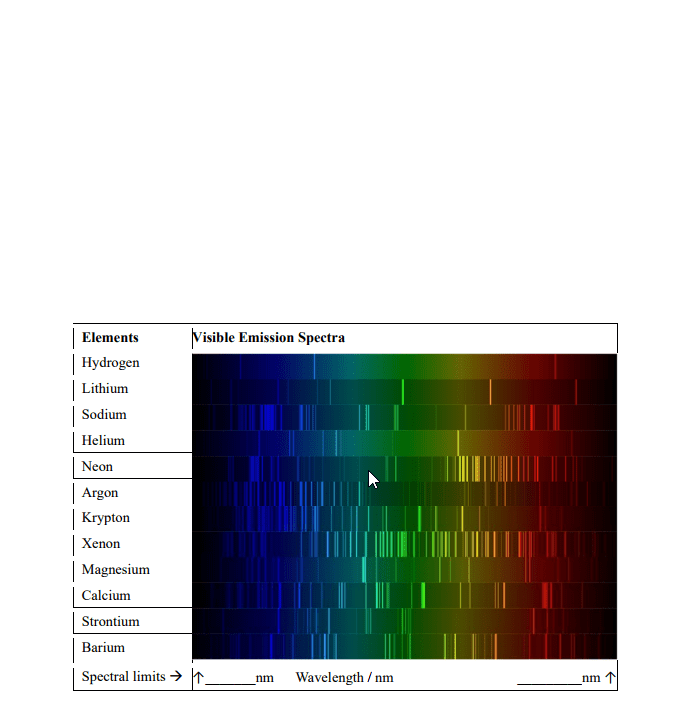
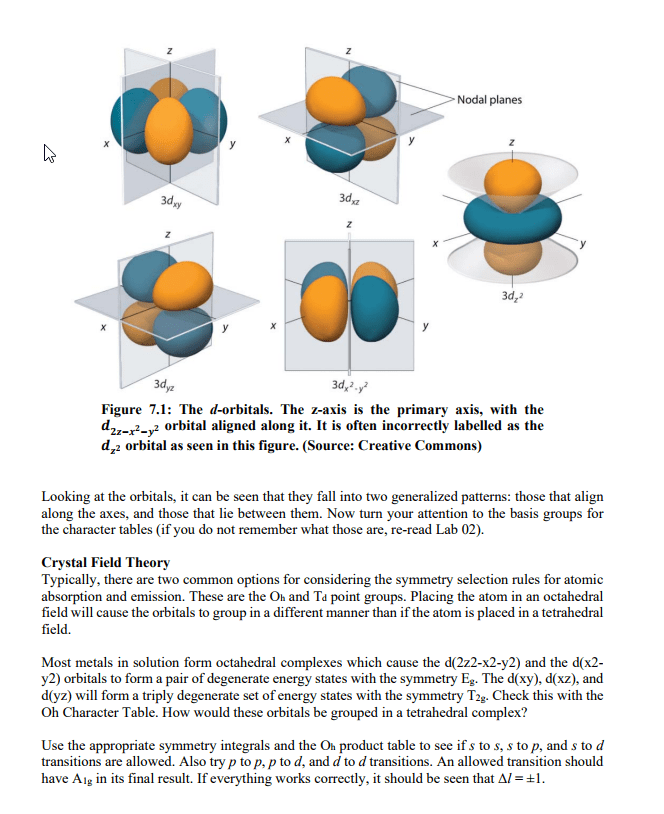
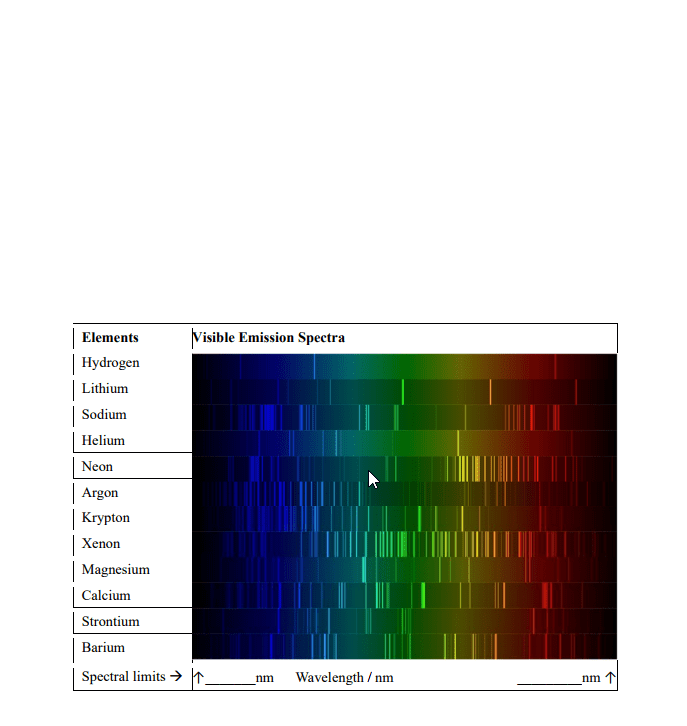
Quantized energy: only allowed/orbit in their certain distance from the nucleus. If energy is gained or lost in an electron, it can jump to a higher level or fall to a lower level. The electrons still hold distinct energy levels, but not in as much of a cut and dry way. Ground state: the lowest level of energy that an electron can be in possible to jump to higher states of energy. Excited state: a new state for an electron once it has gained enough energy and can fall down to lower states (but not necessarily the ground state right away) N = energy level of the electron (1, 2, 3, etc). Finding the differences in energy would include subtracting this equation for one energy level from another. Examples of the subshells and their distinct orbital shapes. It is important to know s and p.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232