CHEM 11200 Lecture Notes - Lecture 19: Rate-Determining Step, Reaction Rate, Activation Energy
28 views1 pages
19 Jul 2020
School
Department
Course
Professor
Document Summary
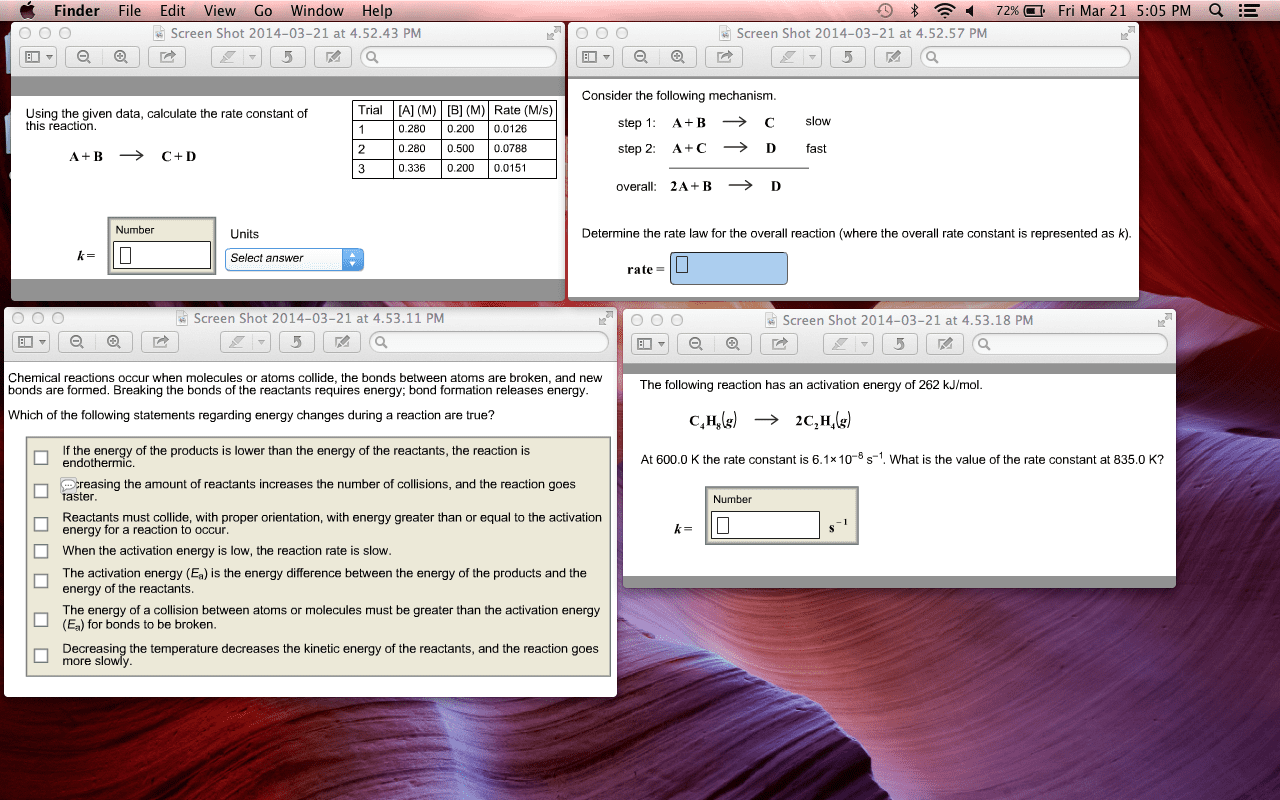
Reaction rate: the rate of a reaction indicates how fast reactants are consumed or how fast products are being formed. Activation energy == the minimum energy required for the reaction to go from reactant to product. When the reactants posses the proper activation energy, they can hit the transition state. Lower the activation energy = faster reaction rate. Greater concentration of reactants = faster reaction rate. Increase the speed of reaction by lowering the activation energy which will. Remains unchanged at the end of a reaction. X and y = the order of reaction. Unimolecular energy = 1st order means that when conc is doubled, reaction rate is doubled.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232