CHEM 001 Lecture Notes - Lecture 22: Hydronium, Decimal Mark, Partial Pressure
Document Summary
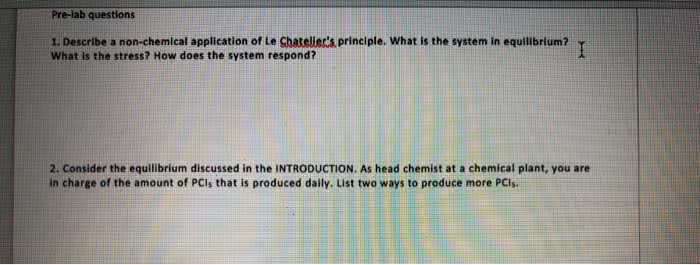
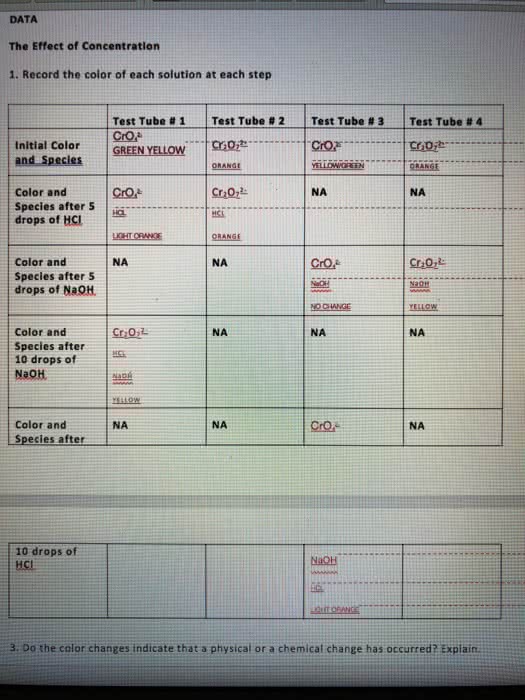
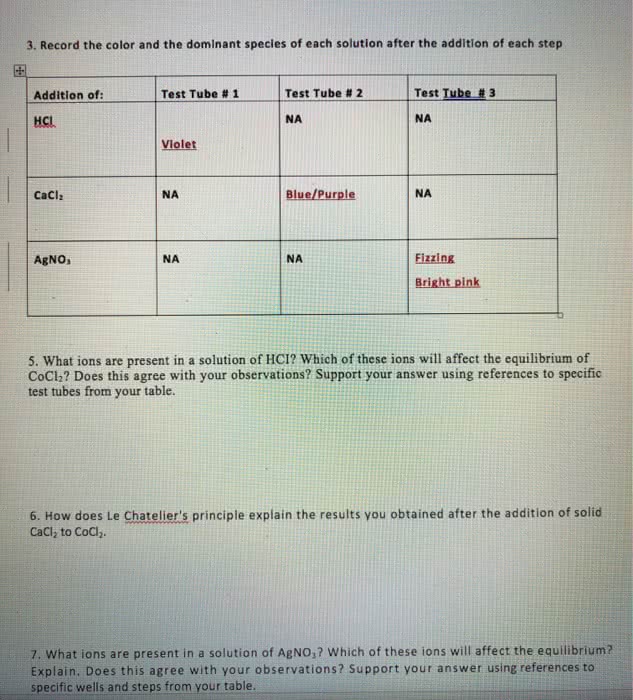
Le chatelier"s principle states that when a system in chemical equilibrium is disturbed by a change in temperature, pressure, or concentration, the system shifts in equilibrium in a way that tends to counteract the change. One can increase or decrease the yield of products through: changing the concentration by removing or adding products or reactants to the reaction vessel, changing the partial pressure of gaseous reactant and products, changing the temperature. Once we have determined the equilibrium constant for a reaction, we can use it to calculate the concentrations of substances in an equilibrium. Chapter 15: 15. 27, 15. 28, 15. 29, 15. 31, 15. 33, 15. 35, 15. 51, 15. 53, 15. 57, 15. 59, 15. 61, 15. 67, Chapter 16: 16. 1, 16. 9, 16. 23, 16. 25, 16. 35, 16. 39, 16. 41, 16. 45, 16. 51, 16. 53, 16. 55, 16. 57, 16. 59, 16. 63, 16. 65, 16. 101, 16. 115 ph, poh pk p (anything) = -log10 (anything) Simply produces an ordinary looking number from a scientific number. For logarithms, sig figs appear as digits after the decimal point.