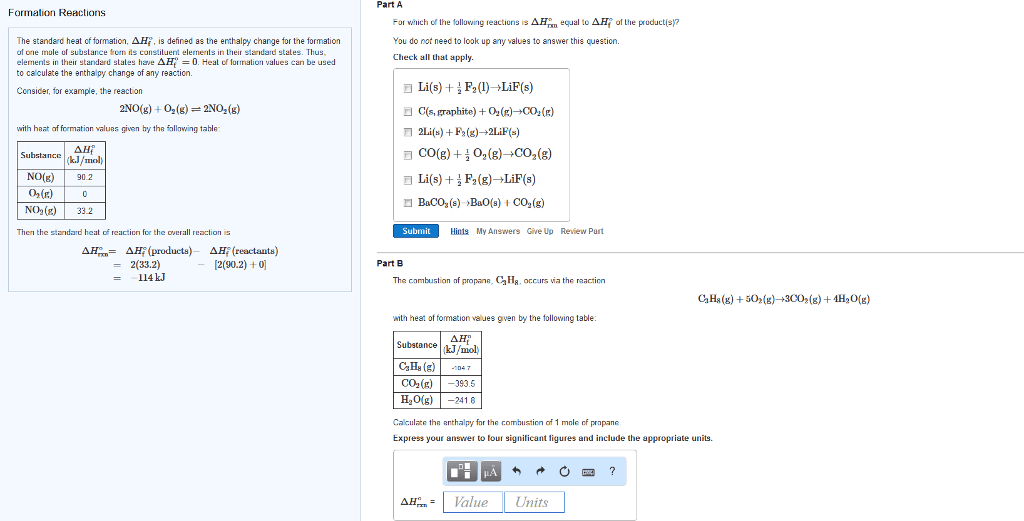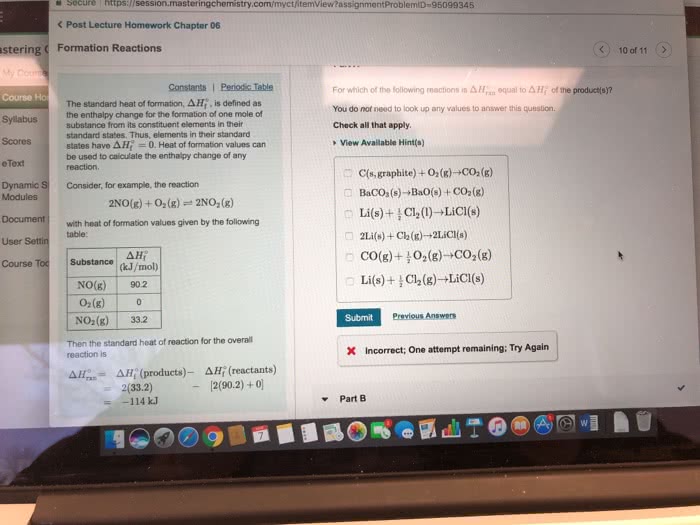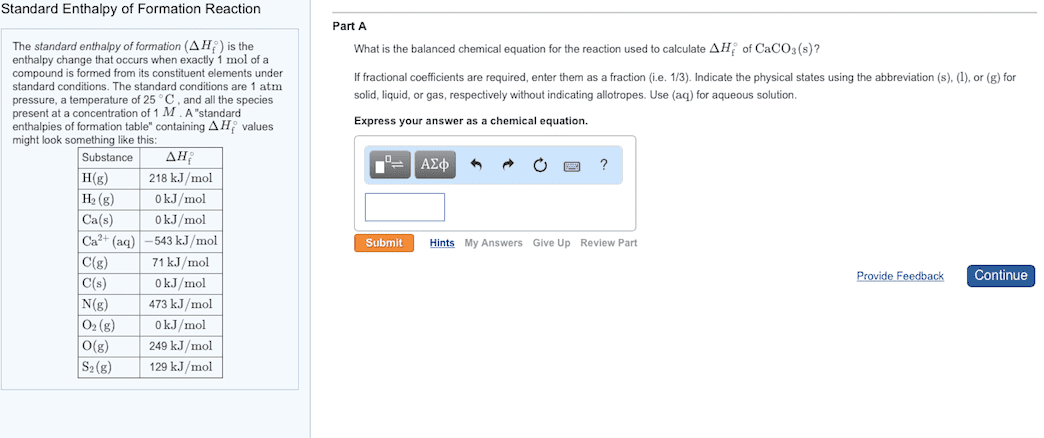
Standard Enthalpy of Formation Reaction The standard enthalpy of formation (delta H f ) is the enthalpy change that occurs when exactly 1 mol of a compound is formed from its constituent elements under standard conditions. The standard conditions are 1 atm pressure, a temperature of 25 degree C , and all the species present at a concentration of 1 M . A standard enthalpies of formation table containing Hf values might look like this:Part A What is the balanced chemical equation for the reaction used to calculate delta Hf of CaCO3(s)? If fractional coefficients are required, enter them as a fraction (i.e. 1/3). Indicate the physical states using the abbreviation (s), (1), or (g) for solid, liquid, or gas, respectively without indicating allotropes. Use (aq) for aqueous solution. Express your answer as a chemical equation.