CHEM 1210 Lecture Notes - Lecture 8: Conjugate Acid
28 views2 pages
Document Summary
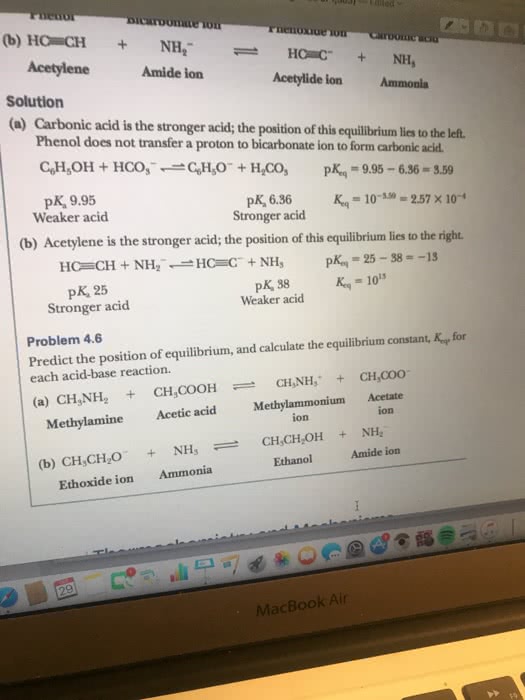
12:17 pm: properties of acids and bases. Acids and bases react with each other in a process called neutralization. The stomach produces hydrochloric acid to break down food and destroy bacteria but is the cause of heartburn. 2hcl (strong acid) + caco3 (strong base) = cacl2 (weak base: acid-base equilibria. We know that hcl is a strong acid, which means that the position of this equilibrium lies very far to the right. In contrast, acetic acid is a weak acid, and the position of its equilibrium lies very far to the left. To predict the position of an acid-base equilibrium such as this, we do the following: Identify the two acids in the equilibrium; one on the left and one on the right. Using the information in table 8-2, determine which is the stronger acid and which is the weaker acid.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232