CHEM 1210 Lecture Notes - Lecture 2: Electron Affinity, Ionic Radius, Diamagnetism
Document Summary
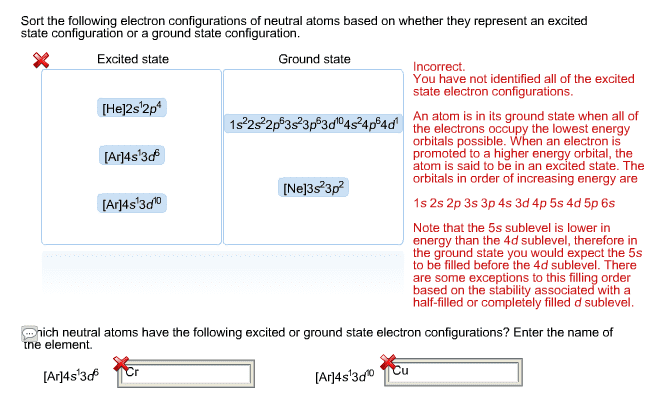
An atom or ion that contains unpaired electrons. An atom or ion in which all electrons are paired. Cations are much smaller than their corresponding neutral atoms. Anions are much larger than their corresponding neutral atoms. The energy required to remove an electron from the atom or ion in the gaseous state. Look at orbital diagrams to see possible exceptions (ex: for second period there is a drop at b and o) Generally becomes more negative (increases) (adding an electron becomes more exothermic) as we move across to the right in the periodic table. Increases down and to the left (bottom left corner very metallic, top right least metallic) The bond that forms between a metal and a nonmetal. The bond that forms between two or more nonmetals. Gives the relative number of atoms of each element in a compound. (simplest ratio) Gives the actual number of atoms in each element in a molecule of a compound. (unsimplified ratio)