METEO 300 Lecture Notes - Lecture 18: Control Volume, Isentropic Process
Document Summary
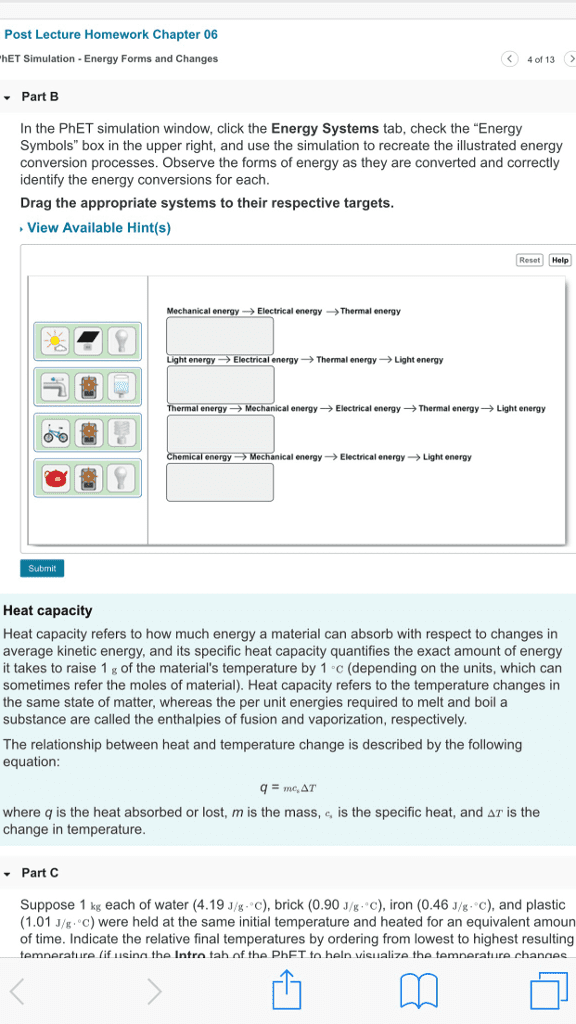
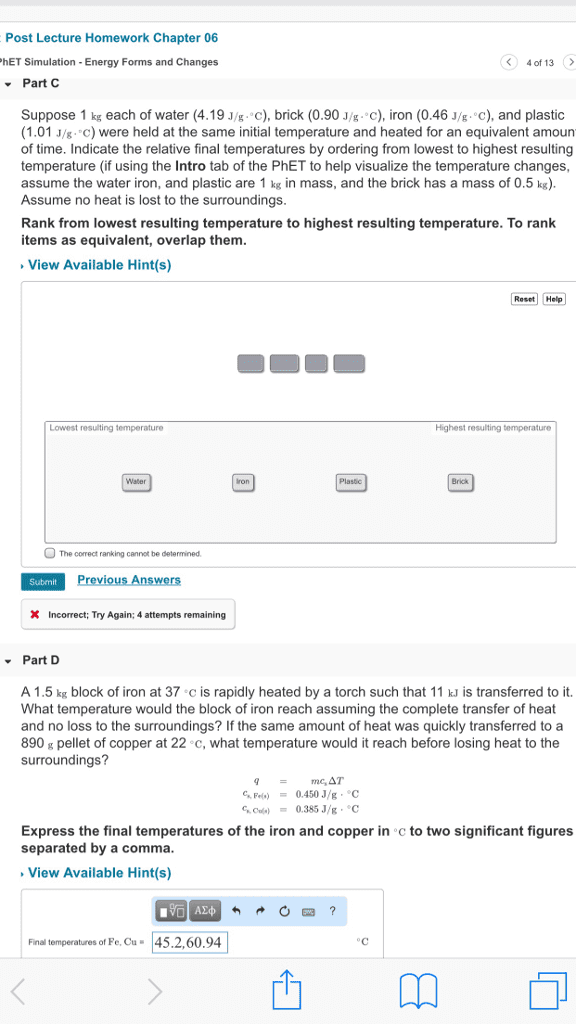
Me 300 - lecture 18 - energy conservation for a control volume. Energy conservation: = where = + + , is mass specific energy and = + + . *the equation for energy conservation of a control volume ultimately becomes: Air (ideal) enters an adiabatic nozzle at 300 k with a steady velocity of 10 m/s. Determine the temperature at the nozzle exit where the velocity is 250 m/s. * = = 0 adiabatic and there is no expansion/compression work. The energy conservation equation becomes: constant constant-pressure specific-heat. *this is an accurate answer because since velocity increases, kinetic energy increases. The change in enthalpy is the negative of the change in kinetic energy, so temperature must decrease. An adiabatic steam (non-ideal) turbine extracts power from a stream of steam steadily flowing at 38. 7 kg/s. At the inlet to the turbine, the steam is at a temperature of. 780 k and a pressure of 10 mpa.