01:160:162 Lecture Notes - Lecture 21: Equilibrium Constant, Lead, Sodium Chloride
Document Summary
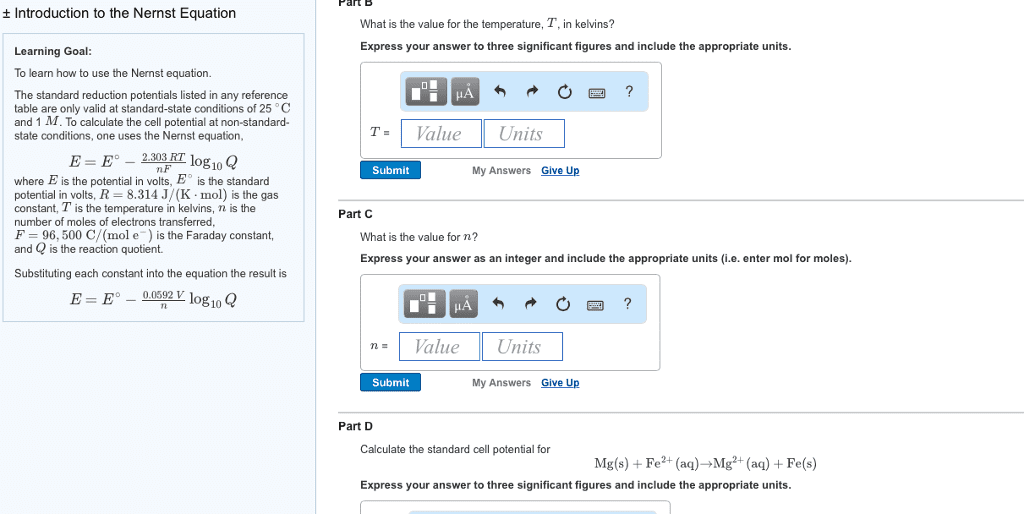
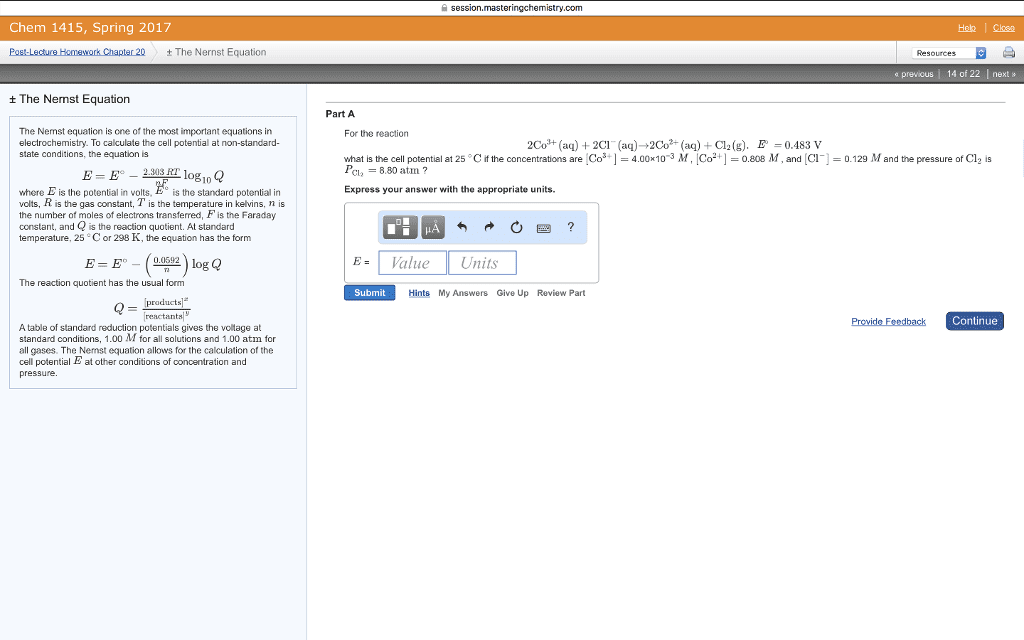
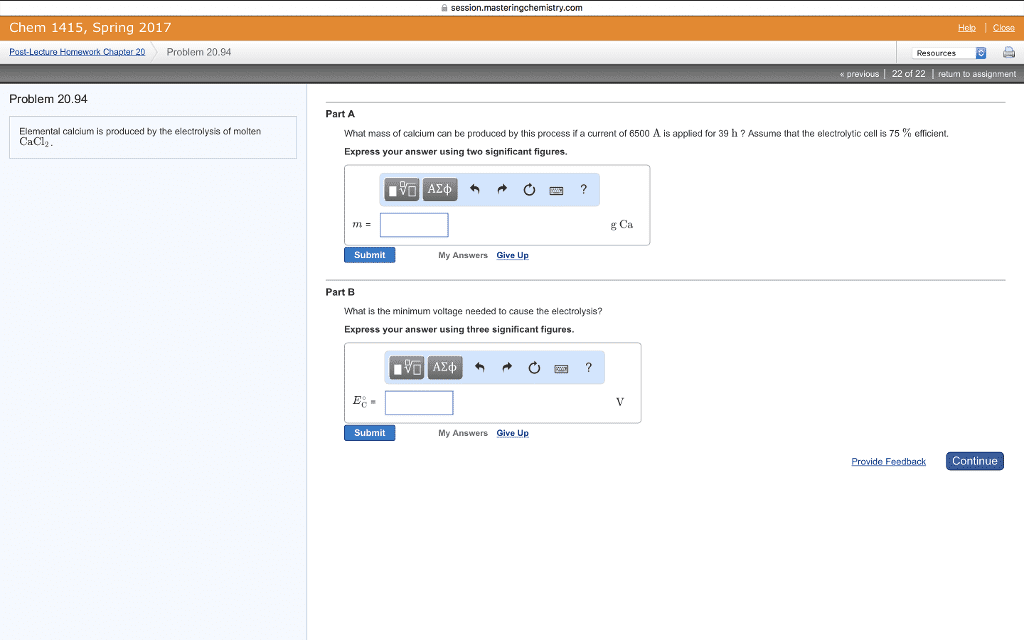
What is the voltage of such a cell if: [mn2+] = 0. 010 m [al3+] = 1. 50 m. What is k for: cu + 2ag+ 2ag + cu2+ log k = At equilibrium: e = 0 and q = k. 025693 n ln k ln k = ne o. What is k for: 2ag + cu2+ cu + 2ag+ log k = A concentration cell is one which generates a voltage based on differences of concentration. In the diagram below, both electrodes are silver, but there are different ag+ concentrations for the anode and cathode. anode cathode. Cathode: ag+ (1. 0 m) + e- ag(s) Anode: ag(s) ag+ (0. 10 m) + e- Ag(s) + ag+(1. 0 m) ag+(0. 10 m) + ag(s) E = 0 - 0. 0592 log (0. 10) e =0. 0592volts anode cathode. Ag(s) + ag+(1. 0 m) ag+(0. 010 m) + ag(s) E = 0 - 0. 0592 log (0. 010) e = 0. 118 volts. Anode: ag(s) ag+(1. 6 x 10-10m) + e-