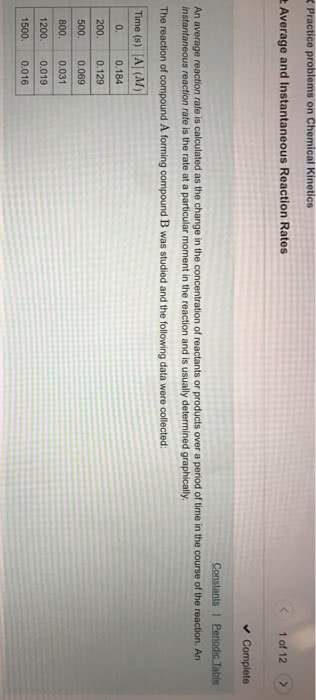
For a reaction aA rightarrow Products, [A]_o = 13 M, and the first two half-lives are 35 and 70 minutes, respectively, a) Write the rate law for this reaction. b) Calculate k. c) Calculate [A] at t = 335 minutes The Model of Chemical Kinetics represents the reaction rate constant as: k = (2p) times exp(-Ea/RT). What does each variable in the equation mean- that is, in regard to their contribution towards the reaction rate constant? In other words, what does each variable actually mean/refer to? Chemists commonly use a rule of thumb that an increase of 10 K in temperature doubles the rate of a reaction. What must the activation energy be for this statement to be true for a temperature increase from 63 degree C to 73 degree C? ln(k_2/k_1) = E_a/R(1/T_1 - 1/T_2) (i) What is a catalyst? (ii) What are the two types of catalysts? Explain specifically why they are different? (iii) What is one example of each type of catalyst? (i) What does it mean when a reaction is in chemical equilibrium? Specifically, within the context of reactants? products, and the forward and reverse reaction rates, what does chemical equilibrium mean? (ii) What is Le Chateleir's Principle? Within the context of reactant/product concentration, temperature, and pressure, how does Le Chatelier's Principle predict a reaction will respond to variations in reactant, product concentration, temperature, and pressure. (i) For the given reactions: N_2(g) + 3H_2(g) harr 2NH_3(g), H_2O(g) + CO(g) harr H_2(g) + CO_2(g), HCN(aq) harr H^+(aq) + CN^-(aq), 2KClO3(s) harr 2KCl(s) + 3O_2(g) and 2H_2 O(l) harr 2H_2(g) + O_2(g). Write the equation that governs each of their equilibrium rate constant K (ii) If N_2(g) + 3H_2(g) harr 2NH_3(g)'s (at 25 degree C) has K = 25, what would K be (at 25 degree C) for 2NH_3(g) harr N_2(g) + 3H_2(g)? (iii) What would K be if [5 times (N_2(g)+3H_2(g) harr 2NH_3(g)]? (i) For N_2(g) + 3H_2(g) harr 2NH_3(g): P_NH3: 1.9 times 10^-1 atm, P_N2 = 8.9 times 10^-2 atm, P_H2 = 1.9 times 10^-2 atm, what is K_p? (ii) Using K_p, calculated in the previous step, what is K at 45 degree C, where K_p = K(RT)^Delta n? (R = 0.08206 L atm/mol K). What is the difference between homogeneous and heterogeneous? (ii) If K is larger than 1, does the equilibrium life farther to the right (favoring the production of products) or to the left (favoring reactant formation)? (iii) If K is less than 1, does the equilibrium life farther to the right (favoring the production of products) or to the left (favoring the production of reactants)?