CHE 201LLR Lecture Notes - Lecture 5: Hydroboration, Carbon–Carbon Bond, Boron
Document Summary
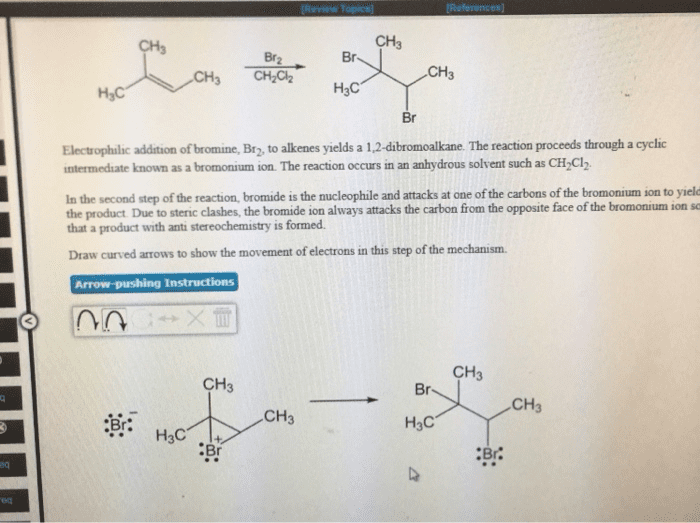
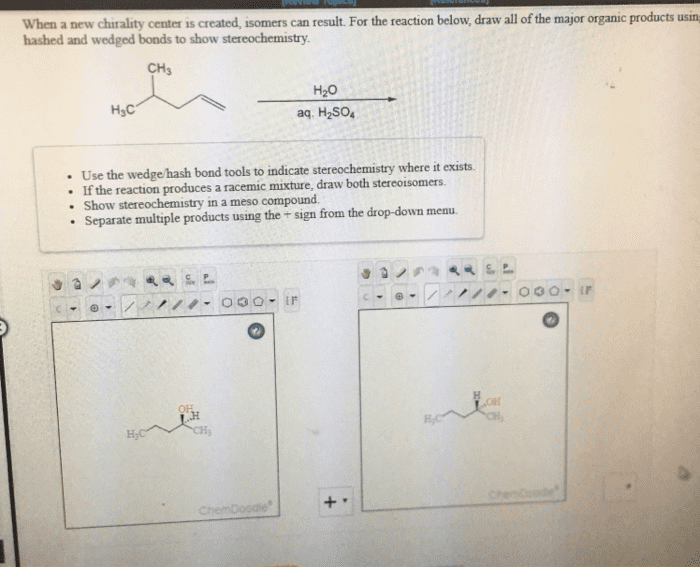
Addition of the halogen creates vicinal dihalides. The eletrons attack the bromine as the bromine attacks the carbon of the double bond. Bromide ion then attacks a carbon of the bromonium ion. Formed when a nucleophilic solvent is used. The solve is present in a large excess. High regioselectivity observed when one carbon disubstituted. 90% of the positive charge resides on the tertiary carbon. C-br bond is so long and weak it is essentially a carbocation. Solvents are generally written under the arrow. Reactants and catalysts are written over the arrow. Have to worry about rearrangements of the carbocation. Reduction step: the h comes from the borohydride. Rearrangements do not occur since there is no carbocation. Boron ((cid:2871)(cid:4667) adds -h and - (cid:2870) Oxidation replaces boron with an regioselectively to alkene alcohol. Alcohol ends up bonded to the carbon with fewer alkyl substituents (anti-markovnikov) Hydroboration step: boron becomes bonded to the carbon with fewer alkyl substituents.