CHEM 134 Lecture Notes - Lecture 6: Chemical Kinetics, Rate Equation, Reaction Rate
22 views4 pages
5 Feb 2018
School
Department
Course
Professor
Document Summary
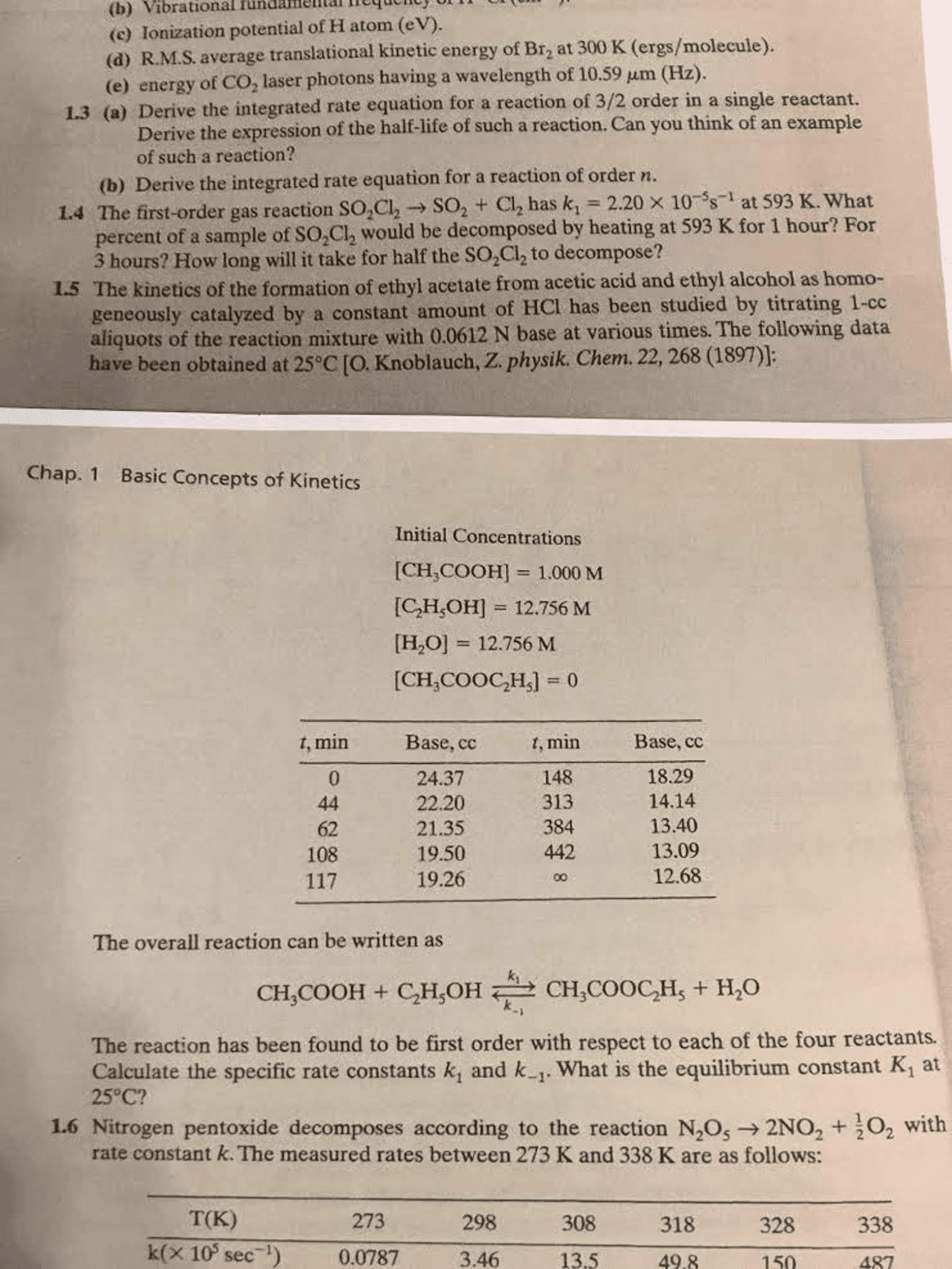
Study of reaction rates, or the changes in the concentrations of reactants and products with time. By studying kinetics, insights are gained into how to control reaction conditions to achieve a desired outcome, its mechanism. Chemical kinetics of a reaction depend on various factors. Reaction rate = change in concentration of a reactant or product with time. Consider the hypothetical reaction, a(g) b(g) Equimolar amounts of reactant a will be consumed while product b will be formed as indicated in this graph: Mathematically, the rate of a reaction can be written as: Expresses the rate of a reaction in terms of changes in the concentration of one or more reactants, [r], over a specific time interval, t. Describes what is occurring on a molecular level during a reaction. Describes the rate of a reaction in terms of the initial concentration, [r]0. , and the measured concentration of one or more reactants, [r], after a given amount of time, t.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232