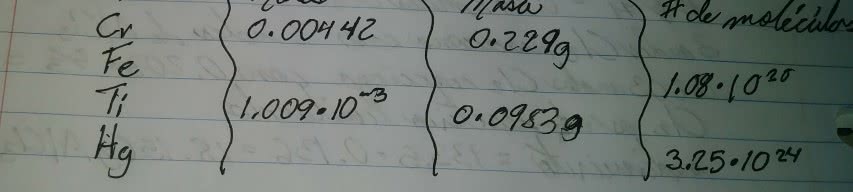CHM 111 Lecture Notes - Lecture 13: Molar Concentration, Stoichiometry, To-3


26
CHM 111 Full Course Notes
Verified Note
26 documents
Document Summary
Chemical reactions in which the reactants are dissolved in water are among the most common and important. For example, the reactions that occur in lakes, streams, and oceans, as well as the reactions that occur in every cell of our bodies, take place in water. A homogeneous mixture of two substances, such as salt and water, is called a solution. The majority component of the mixture is called the solvent, and the minority component is called the solute. Moreover, an aqueous solution is a solution in which water acts as the solvent. The amount of solute in a solution is variable. A common way to express solution concentration is molarity (m), the amount of solvent (in mol) divided by the volume of solution (in liters). Molarity (m) = amount of solute (in mol [ ] = brackets also denote molarity. Volume of solution (in l) m = m also indicates molarity.