CHEM 1C Lecture Notes - Lecture 4: Hydroxylamine, Reaction Mechanism, Molecularity

➔Low activation barrier → higher rate of reaction
➔
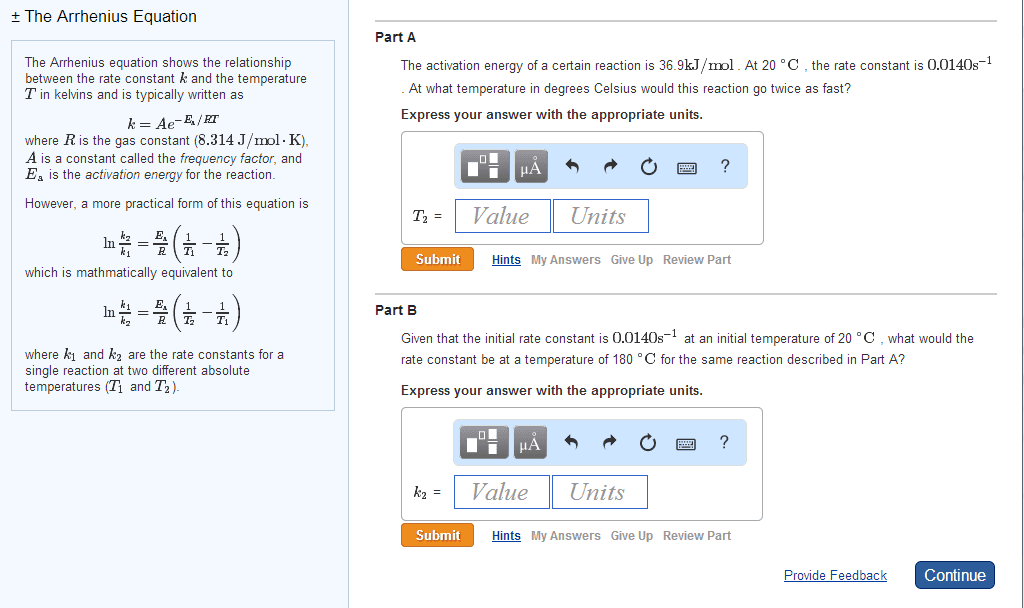
➔Reaction rate constants generally change with temperature
➔Many reaction rates constants are empirically known to follow Arrhenius law:
◆
◆Ea is the activation energy (J/mol) ln(k) has a linear dependence on 1/T
◆R = NakB = 8.314 J/Kxmol slope: -Ea/R Intercept: ln(A)
◆T is the absolute temperature
◆A is the pre-exponential factor
Example: Cyclopropane isomerizes to propylene in a first-order reaction. The activation energy
is 272 kJ/mol. At 500℃ the rate constant is 6.1 x 10^-4 1/s. Calculate the Arrhenius prefactor, A,
for the reaction and the rate constant at 25℃.
Document Summary
Low activation barrier higher rate of reaction. Reaction rate constants generally change with temperature. Many reaction rates constants are empirically known to follow arrhenius law: A is the pre-exponential factor ln(k) has a linear dependence on 1/t slope: -ea/r intercept: ln(a) Example: cyclopropane isomerizes to propylene in a first-order reaction. At 500 the rate constant is 6. 1 x 10^-4 1/s. Calculate the arrhenius prefactor, a, for the reaction and the rate constant at 25 . Assuming a and ea do not depend on t, at 298 k: Example: the decomposition of hydroxylamine (nh2oh) in the presence of oxygen follows the rate law: rate = k[nh2oh][o2]. The rate constant k = 0. 237 x 10^-4 1/ms at 0 c and 2. 64 x. The overall progress of a chemical reaction can be represented at the molecular level by a series of simple elementary steps or elementary reactions. The sequence of elementary steps that leads to product formation is the reaction mechanism.