CHEM 001A Lecture Notes - Lecture 10: Molar Attenuation Coefficient, Barium Chloride, Molar Mass
59 views2 pages
Document Summary
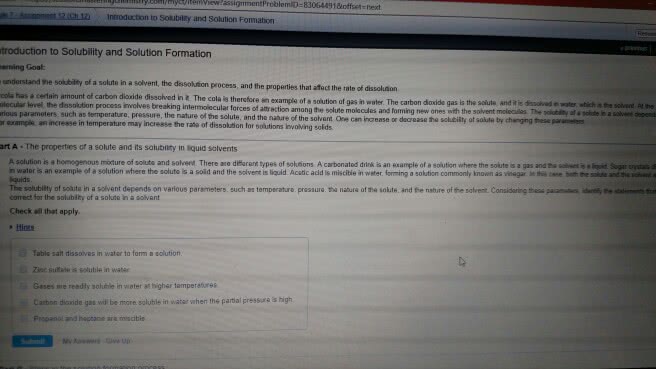
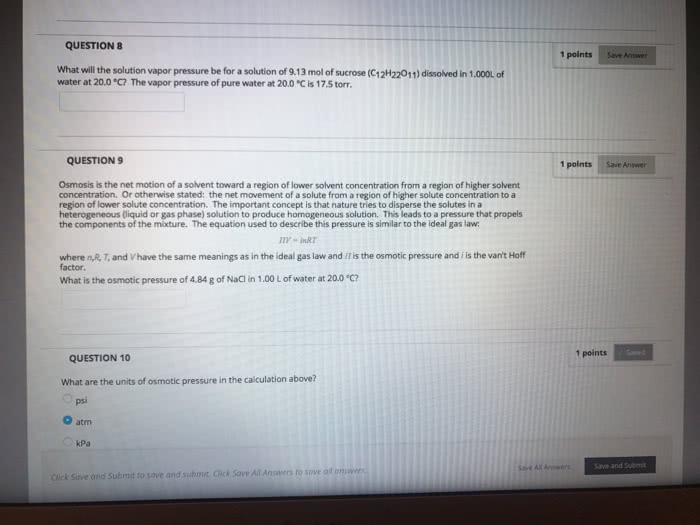
Earth"s surface is about 75 percent covered by water. Properties of water are responsible for life on earth and many geographical. All natural waters have ionic and molecular compounds dissolved in them. Solutions : homogeneous mixtures of two or more substances. A solution may contain one or more solutes. Aqueous solutions water is the solvent. Is the amount of a solute in a particular amount of solvent or amount of solute amount of solvent. Quantitative concentration units based on: amount of solute amount of solution or . Example : concentration of molecules in blood are given in units of mg milligrams of substance (solute) per deciliter: dl. Parts per million ( ppm ): 1 mg solute. Parts per billion ( ppb ): 1 g solute. If we know the volume (v) and molarity ( m ) of a solution we can calculate moles volume (v ) of solution in liters moles (n) of solute. Moles = mass (g) mass (g)
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232