CHEM 14B Lecture Notes - Lecture 6: Spontaneous Process, Reduction Potential, Membrane Potential
Document Summary
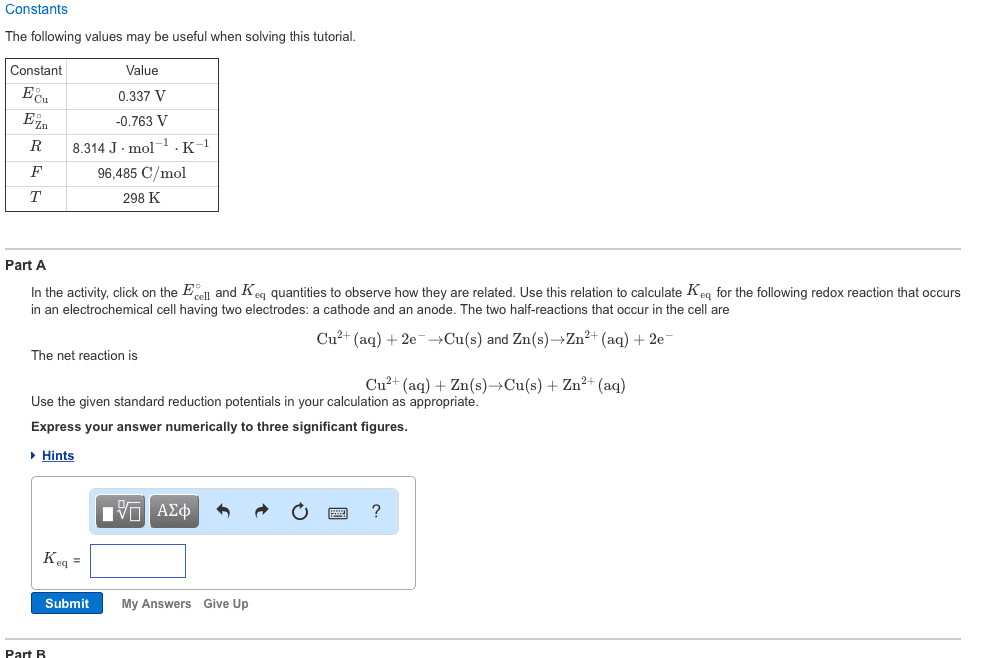
The standard reduction potential (e o ) gives voltage difference between two standard electrodes, which is always the same. The voltage difference is the same and doesn"t depned on how many times the reaction occurs. So, e o stays the same when the half reaction is balanced. One of the half reactions must be reversed to change the sign of the e o. To balance redox reactions, identify the half reactions and look up the standard reduction potentials (e o ) reversed). reaction is balanced, is intensive value). Fe 3+ + e - fe 2+ e p = 0. 77v. Cu 2+ + 2e - cu e o = 0. 34v. Then, balance half reactions and write the cell reaction. Cathode: 2fe 3+ + 2e - 2fe 2+ e o = 0. 77v (e o doesn"t change when the. Anode: cu cu 2+ + 2e - e o = -0. 34v (sign changes because reaction is.