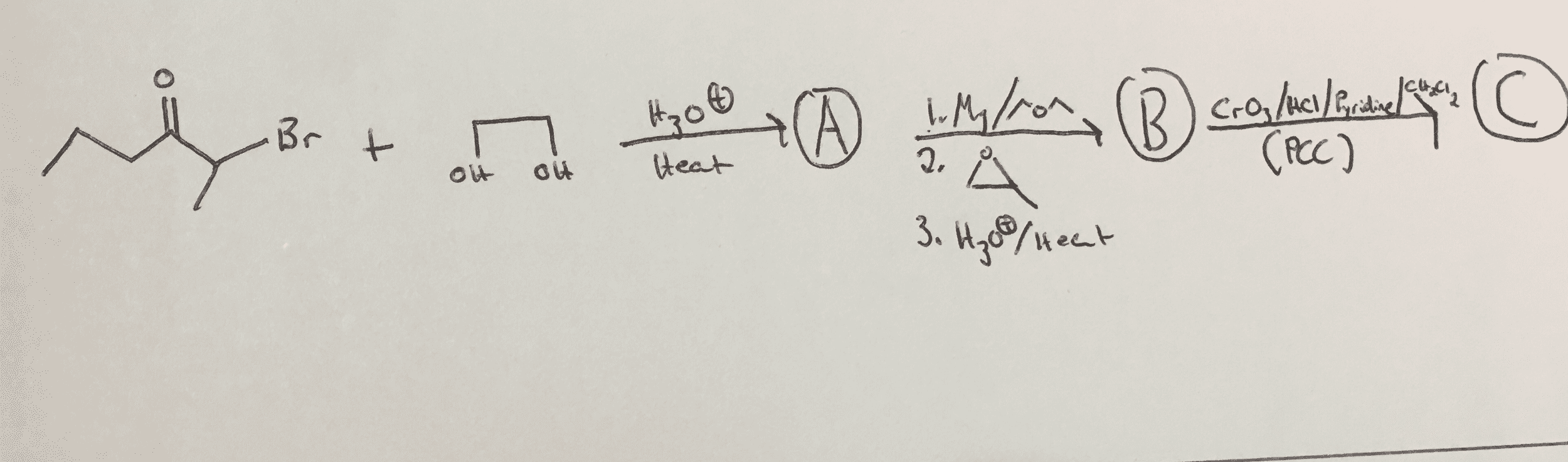CHEM 30A Lecture 5: [Wk2.Wed] Conformational Isomerism
47 views1 pages
20 Oct 2016
School
Department
Course
Professor
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
2. Just like chemical reactions, protein conformational changes are bidirectional. Motor proteins are able to move in one direction by making coordinated conformational changes through the use of ATP. Why does this allow unidirectional movement instead of random walking?
| A. ATP hydrolysis releases a charged phosphate which is used to push the motor protein. | |
| B. The negatively charged phosphates of ATP electrostatically pull the motor proteins. | |
| C. Coupling ATP hydrolysis to one conformational change means that the reverse conformational change would require a large energy input. | |
| D. Coupling ATP hydrolysis to the +âG conformational changes allows the reaction to proceed. |