CHEM 130 Lecture Notes - Lecture 35: Equilibrium Constant, Reaction Quotient, Reagent
Document Summary
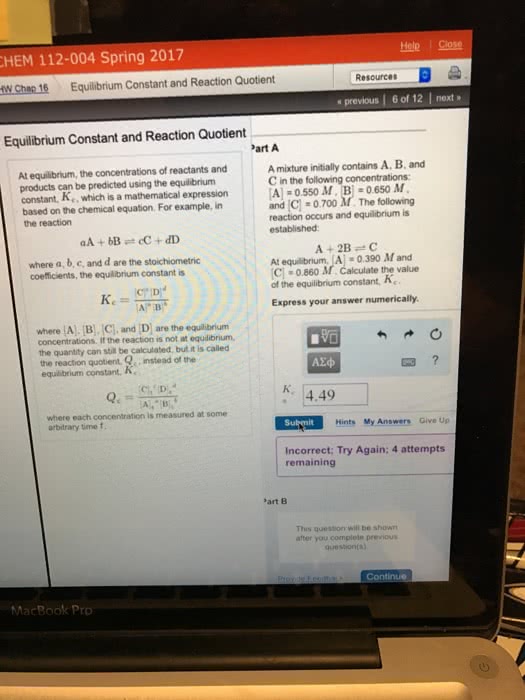
Chem 130: general chemistry: macroscopic investigations and reaction principles - If the value of the equilibrium constant is . Large, the products are favored at equilibrium. Small, the reactions are favored at equilibrium. Neither large nor small (around 1), neither reactants nor products are strongly favored. The equilibrium mixture contains significant amounts of all substances in. If kc for a reaction aa + bb cc + dd is the reaction. Large, the equilibrium mixture is mostly products. Small, the equilibrium mixture is mostly reactants. Around 1, the equilibrium mixture contains appreciable amounts of both reactants and products. An expression that has the same form as the equilibrium-constant expression but whose concentration values are not necessarily those at equilibrium. For catalytic methanation, the reaction quotient is qc = ([ch4]i[h2o]i)/([co]i[h2]i) Subscript i indicates concentrations at a particular instant i. For the general reaction aa + bb cc + dd, q c = ([c]c. If qc > kc i[d]d i)/([a]a i[b]b i), then.