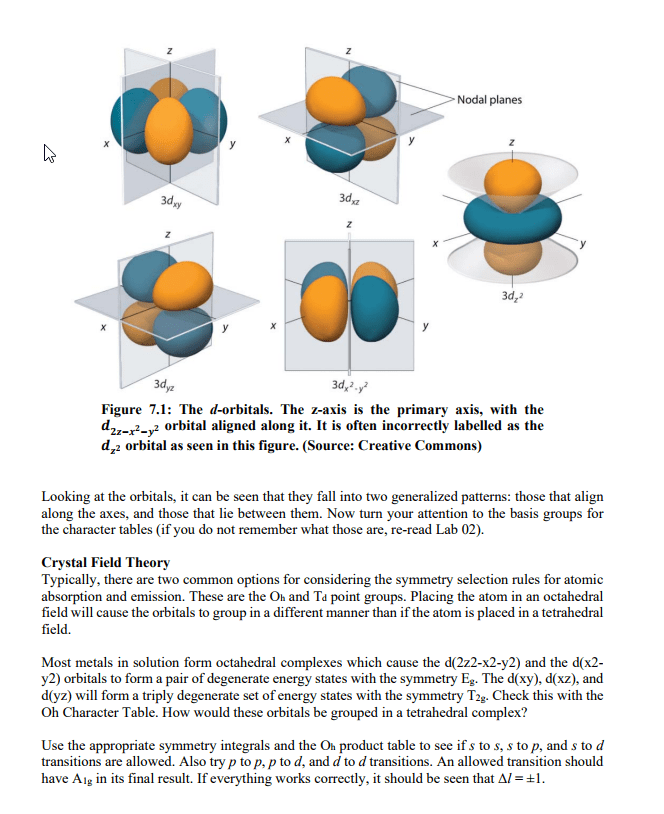
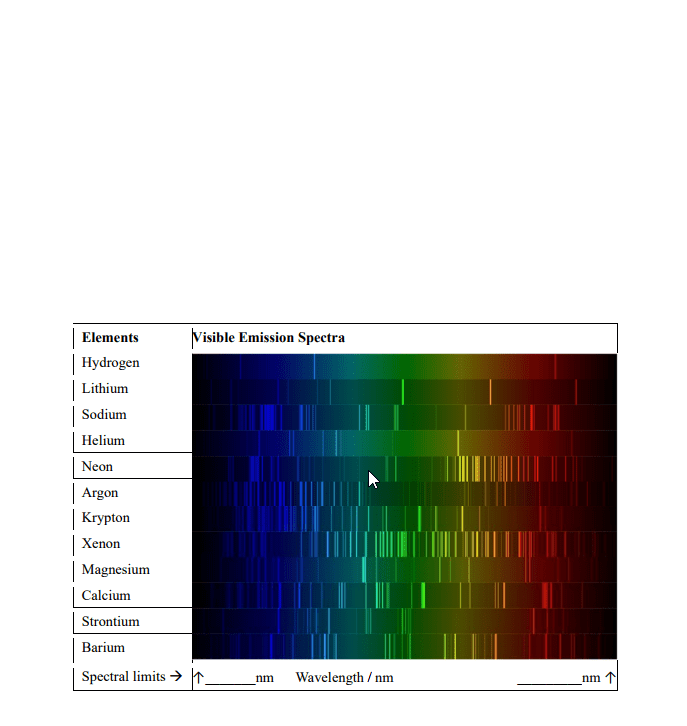
CHEM 130 Lecture Notes - Lecture 19: Emission Spectrum, Continuous Spectrum, Matter Wave
27 views2 pages
Document Summary
Chem 130: general chemistry: macroscopic investigations and reaction principles - Lecture 19: the bohr theory & quantum mechanics. A spectrum containing light of all wavelengths, like the light of a rainbow. A spectrum showing only certain colors or specific wavelengths of light. Bohr established postulates to account for the stability of the hydrogen atom and the line spectrum of the atom. An electron can have only specific energy values in an atom, called its energy levels. Therefore the atom itself can have only specific total energy levels. Energy levels of the electron in the hydrogen atom can be derived by e = An electron in an atom can change energy only by going from one energy level to another energy level. By doing so, the electron undergoes a transition. The energy of the emitted photon, hv, equals the positive energy lost by the atom (- e) Hv = - e = -(ef - ei)
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232