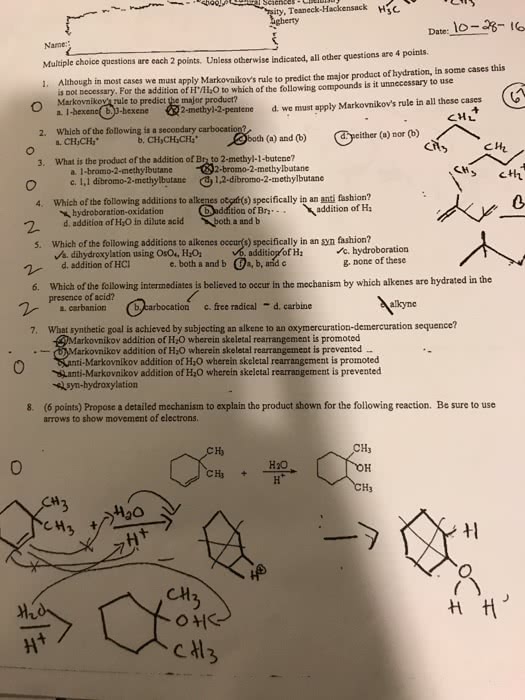
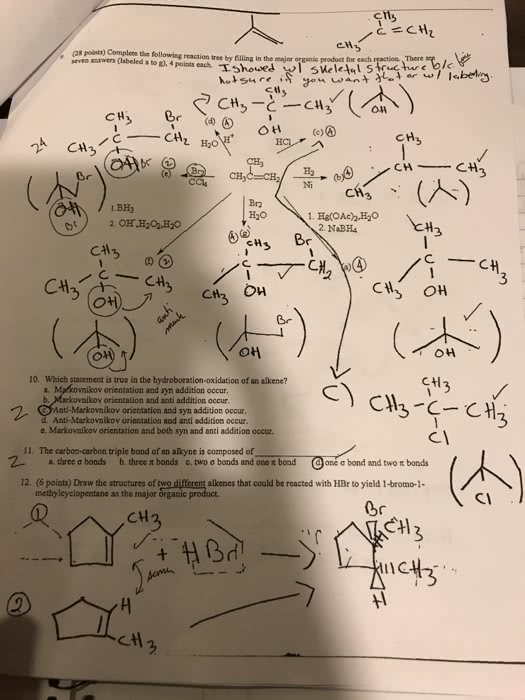
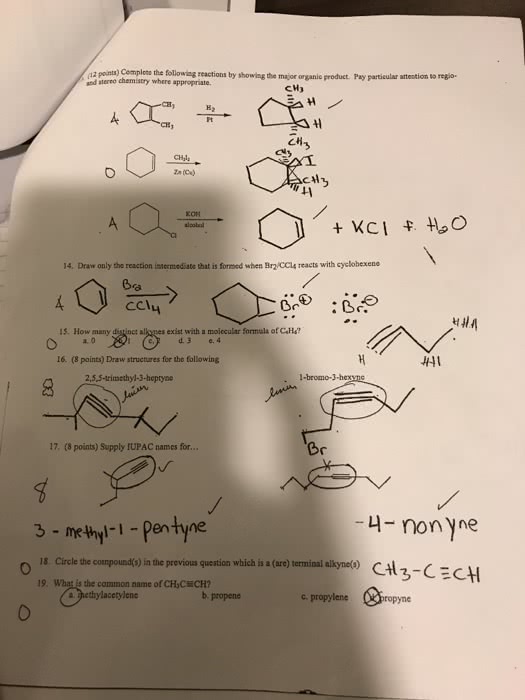
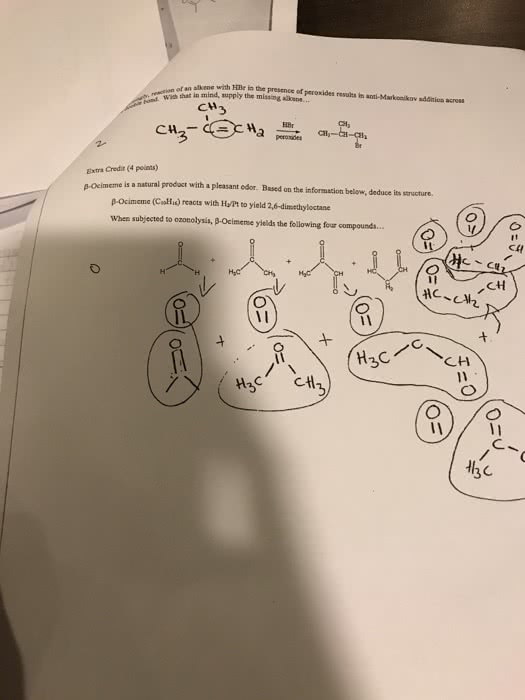
How do you solve #8?

Brooklyn College of C.U.N.Y Spring 209 Chemistry 1.2 Makeup Midterm Exam Lab. Sectio Physical Constants R-0.0821 L.atrn /mM1 K: 1 atrm-760 Torr; h-6.626 x 10" Js. c-300 x 10' mis Multiple Choice Question4 points each. Circle the correct choice I. How many orbitals are there in the n - 3 shel 2. Which of the following is not a valid set of four b6 ( 4)12 e) none of the preceding choices quantum numbers to describe an electron in an atom? 3. Which pair of elements would you expect to exhibit the most similar physical and chemical peoperties? a) As and Se Na and ci As and Sb s) B and Bensnp 4) The rn unpaired electrons in the ground state of an atom of Co is... ree (C) five (D) seven (E) none of the preceding choices 0è®ä¸ä½å£ wch of the following processes is exothermic? a) lonization of a sodium atom b) Formation of gaseous Which of the following bonds is the most polar? a) N-O b)o-F ) B-o )so e)all are equally polar t what temperature (in C) will 25.0 g of carbon dioxide (at 1.00 atm) occupy 21.3 L? 8°C b) 461 C c) -263°C d)-270°C e) 113°C How many times more rapidly will hydrogen (H,) gas effuse through a pinhole than will deuterium ( Deuterium is an isotope of hydrogen with a molecular mass of 4.0 g'mole. (A) 2 (B)2 (C)2v2 (D)4 9, Which of the following arrangements from lowest to highest in first ionization energies is correct ? 10. In which of the following compounds obeys the octet rule? a) XeF b) BF, c) CIO d) SO, pone of the preceding choicees 11. The bright spectral lines observed in the emission spectrum of a flame or gas discharge tube are due to a) the absorption of light at specific wavelengths b) the formation of an excited state of atoms the transition of an atom from an excited state to a lower energy state d) the change from anti-parallel to parallel electron spins in an atom oxygen at 25 °C. Comparing the pressure of the hydrogen to the exygen, the ratio will be From Bond Dissociation Enthalpies: Î11D (CI-CI)-243 klinolalo (F-F )-159 kJ/mol; Î1lo (CHF)-25 12.A one-liter container is filled with one mole of hydrogen at 25 °C.A second one-liter container is filled with or 2CIF(R) alculate the enthalpy, AH, for the chemical reaction: Cl (g)+ F: (g) (a)-147 kJ (b)-108 kJ (c) +171 kJ (d) +912J (H lou, mory t,hes more rapidy wil, hydrogen w many tes more ro