CHEM 116 Lecture Notes - Lecture 1: Boiling Point, Boiling-Point Elevation, Formula Unit
60 views3 pages
Document Summary
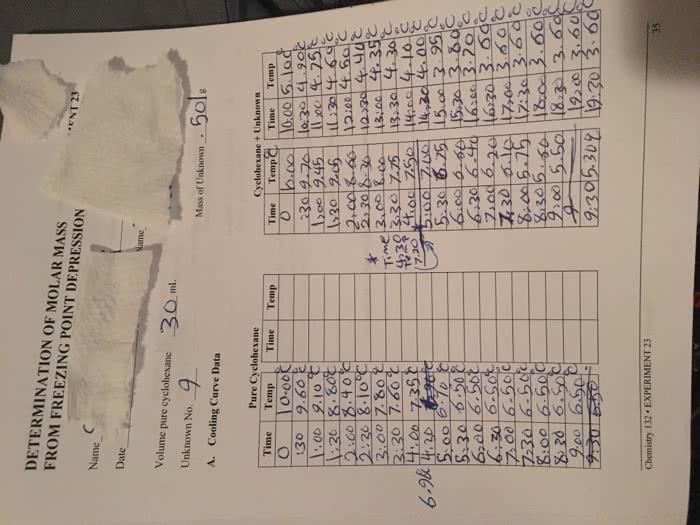
When the rate of exit = the rate of entry, vapor is at equilibrium with the liquid. Vp is smaller in liquid with high imf1s. When vp = atmosphere pressure, liquid boils. Non-volatile solute presence of solute lowers vp zero vp blocks exit of solvent molecules from the surface, lowers the vp. Vp of solvent based on mole fraction of solvent. Psolvent = vp of solvent above solution. Volatile solute solute and solvent have vp. 2 components solute with volatile species a and b both a and b obey raoult"s law. Vap * ptotal boiling point elevation/freezing point depression phase diagram of pure substance shows the phase of the substance at a given pressure and temperature. Symbol the lines are called boundaries and on these boundaries the substance exists in two phases the line from the triple point to the critical point corresponds to vp of the substances as a function of temperature.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232