CAS CH 203 Study Guide - Quiz Guide: Valence Electron, Methylene Group, Formal Charge
Document Summary
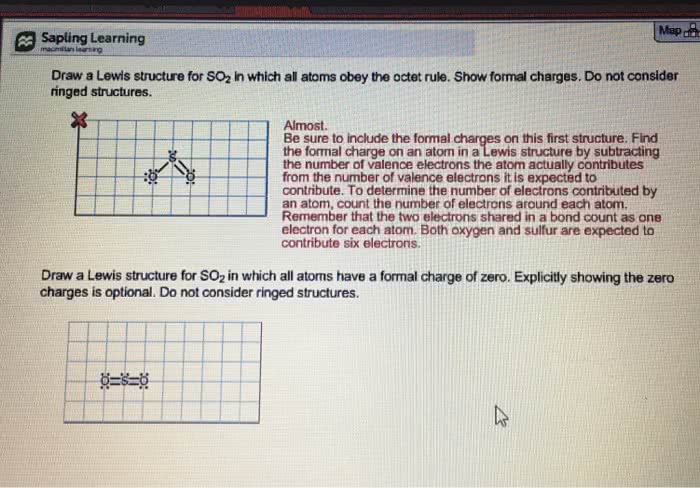
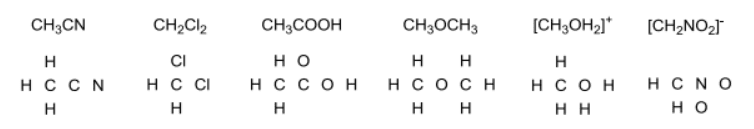
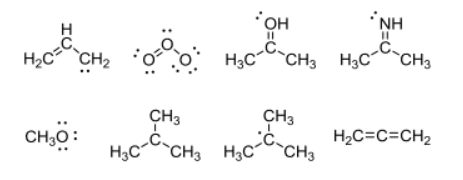
:o: covalent distribution of electrons amongst atoms in a structure. Identify the number of actual electrons around an atom in a structure by drawing a lasso around the atom. L> formal charges (# of ve) - (# of e- atom has) Fc = ( gye_ ) - ( i : methylene group. Bonds are split in half by the lasso, so only half of the bonding electrons count white lone pairs are fully encompassed by the lasso so all nonbonding electrons count. Most atoms in a structure have a formal charge of 0. 1 actual electron hydrogen (1 valence electron) - ( 1 actual electron) = 0. > carbon in ch and ct} groups or any carbons making four bonds.