1
answer
0
watching
659
views
6 Nov 2019
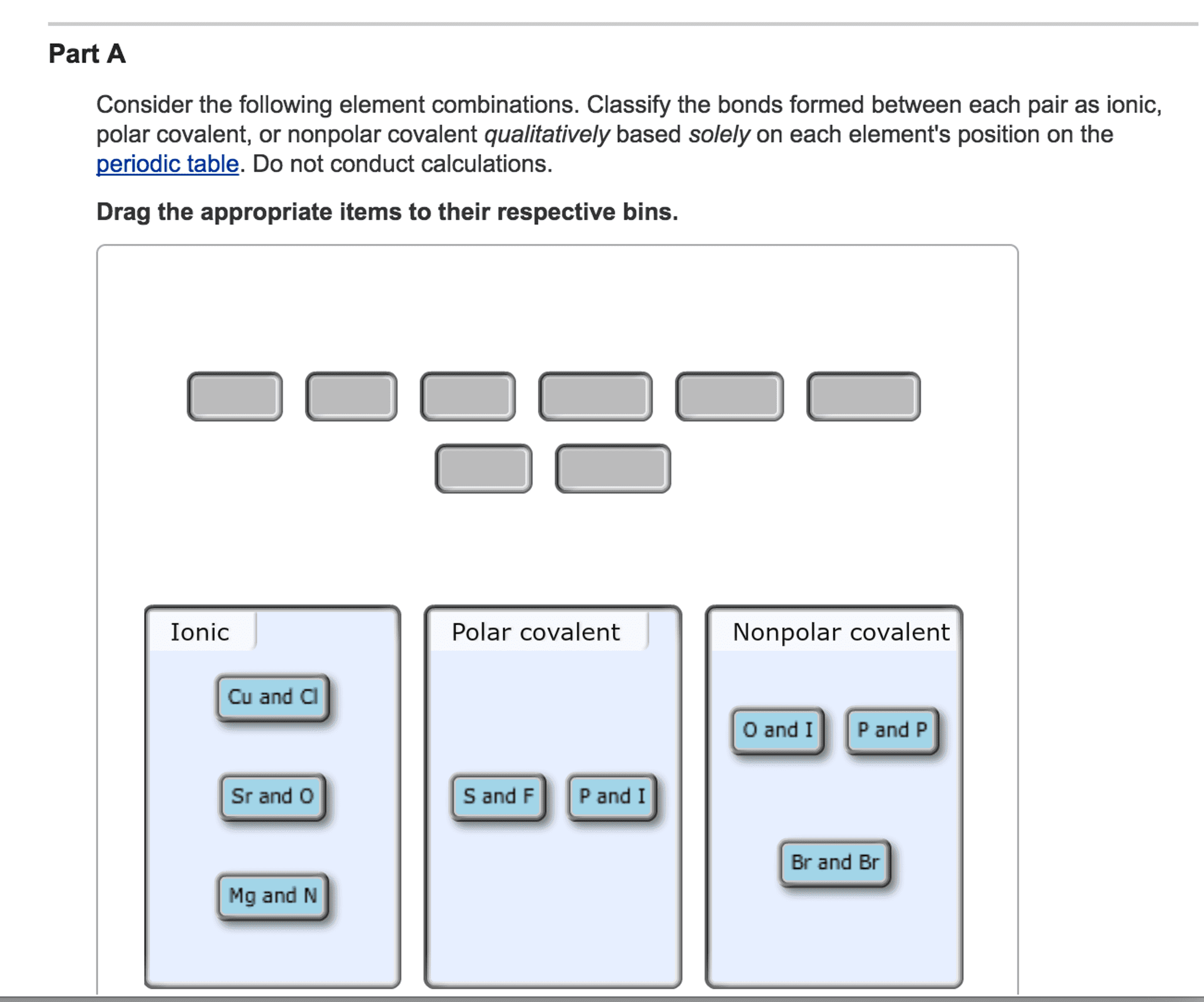
Consider the following element combinations?
i get the next elements:
S-S
Sr-O
Na-N
O-F
S-Cl
I-I
P-Br
Cs-Cl
Classify the bonds formed between each pair as ionic, polarcovalent, or nonpolar covalent based on each element's position inthe periodic table.
Consider the following element combinations?
i get the next elements:
S-S
Sr-O
Na-N
O-F
S-Cl
I-I
P-Br
Cs-Cl
Classify the bonds formed between each pair as ionic, polarcovalent, or nonpolar covalent based on each element's position inthe periodic table.
i get the next elements:
S-S
Sr-O
Na-N
O-F
S-Cl
I-I
P-Br
Cs-Cl
Classify the bonds formed between each pair as ionic, polarcovalent, or nonpolar covalent based on each element's position inthe periodic table.
Jean KeelingLv2
19 Apr 2019