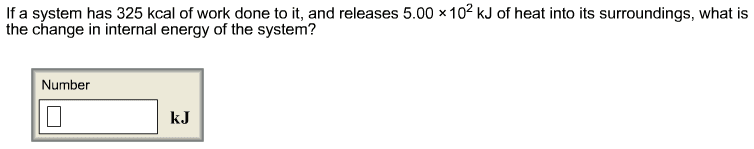Please answer the following questions AND SHOW WORK ifapplicable--I do not understand some of them! (750 value)
1. How many moles of chloride ions are present in 18.4g ofCaCl2?
2. Complete the follow reaction and identify the Bronstedbase:
NaOH (aq) + HCl (aq) ---> ________________
3. A candy bar contains 280 Calories, of which the fat contentaccounts for 120 Calories. What is the energy of the fat content,in kJ?
4. 25 kJ of heat is absorbed and 15 kJ of work is done on thesurroundings. What is /\U?
5. What is the change in internal energy in joules for a systemthat releases 154 J of heat and does 125 J of work on itssurroundings?
6. Ice packs, have lower temperatures when ammonium nitratedissolves in water--which of the following is true of thisreaction?
A. /\H<0, process is exothermic
B. /\H>0, process is exothermic
C. /\H<0, process is endothermic
D. /\H>0, process is endothermic
E. /\H=0, since cold packs are sealed
7. 2SO2 + O2 ----> 2SO3, /\H degrees rxn = -198kJ/mol, howmuch heat is evolved when 600 g of SO2 is burned?
8. Calculate /\H degree rxn for the following reaction:
2H2O2 ---> 2H2O +O2
*Given that /\H degree f [H2O] = -285.8 kJ/mol and /\H dgree f[H2O2] = 187.6 kJ/mol
9. For the following reaction, write the half-reactions, the netreaction, and identify the oxidizing agent:
H2SO4 + Cd(OH)2 ----> 2H2O + CdSO4
THANK YOU!
Please answer the following questions AND SHOW WORK ifapplicable--I do not understand some of them! (750 value)
1. How many moles of chloride ions are present in 18.4g ofCaCl2?
2. Complete the follow reaction and identify the Bronstedbase:
NaOH (aq) + HCl (aq) ---> ________________
3. A candy bar contains 280 Calories, of which the fat contentaccounts for 120 Calories. What is the energy of the fat content,in kJ?
4. 25 kJ of heat is absorbed and 15 kJ of work is done on thesurroundings. What is /\U?
5. What is the change in internal energy in joules for a systemthat releases 154 J of heat and does 125 J of work on itssurroundings?
6. Ice packs, have lower temperatures when ammonium nitratedissolves in water--which of the following is true of thisreaction?
A. /\H<0, process is exothermic
B. /\H>0, process is exothermic
C. /\H<0, process is endothermic
D. /\H>0, process is endothermic
E. /\H=0, since cold packs are sealed
7. 2SO2 + O2 ----> 2SO3, /\H degrees rxn = -198kJ/mol, howmuch heat is evolved when 600 g of SO2 is burned?
8. Calculate /\H degree rxn for the following reaction:
2H2O2 ---> 2H2O +O2
*Given that /\H degree f [H2O] = -285.8 kJ/mol and /\H dgree f[H2O2] = 187.6 kJ/mol
9. For the following reaction, write the half-reactions, the netreaction, and identify the oxidizing agent:
H2SO4 + Cd(OH)2 ----> 2H2O + CdSO4
THANK YOU!