1
answer
0
watching
138
views
11 Nov 2019
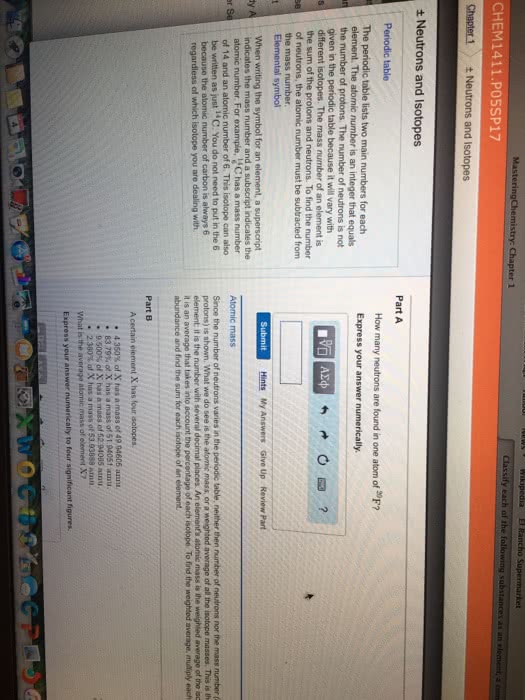
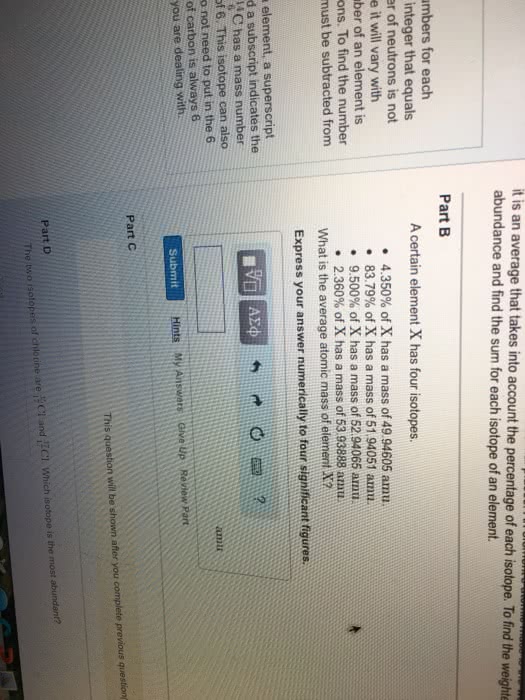
Can you find any other instances in the periodic table where two adjacent elements exhibit a similar behavior, that is, the element with the lower atomic number has a higher atomic mass?
Can you find any other instances in the periodic table where two adjacent elements exhibit a similar behavior, that is, the element with the lower atomic number has a higher atomic mass?
1
answer
0
watching
138
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jamar FerryLv2
13 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
In the periodic table we use today, the ordering of the elements is based on _____.
| triads (groups of three) of elements with similar properties | |
| atomic number | |
| atomic mass | |
| octaves (groups of eight) of elements, which resulted in every eighth element having similar properties |