1
answer
0
watching
181
views
11 Nov 2019
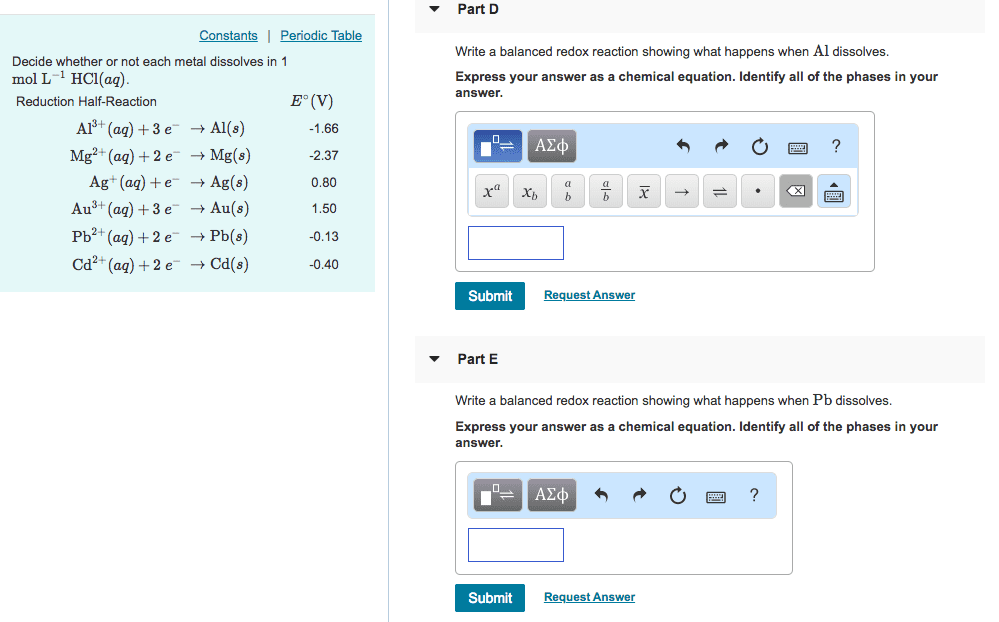
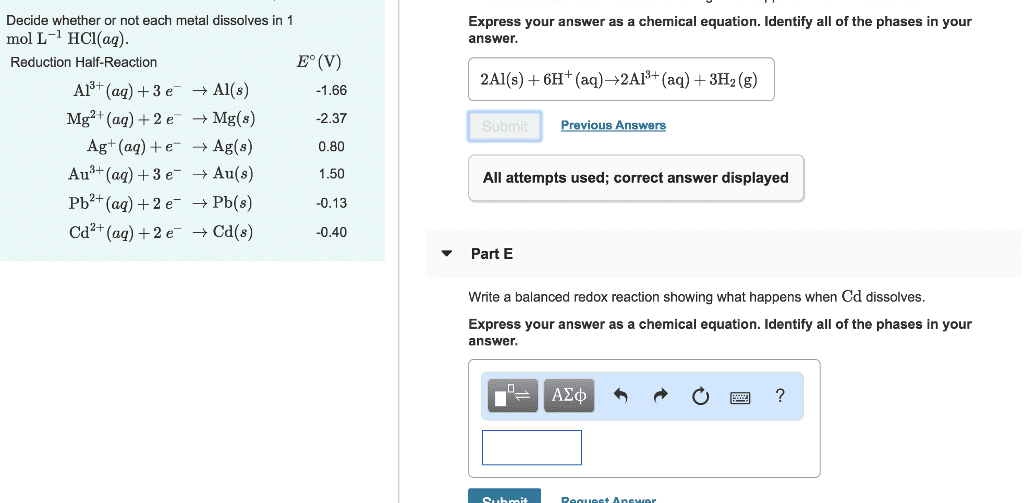
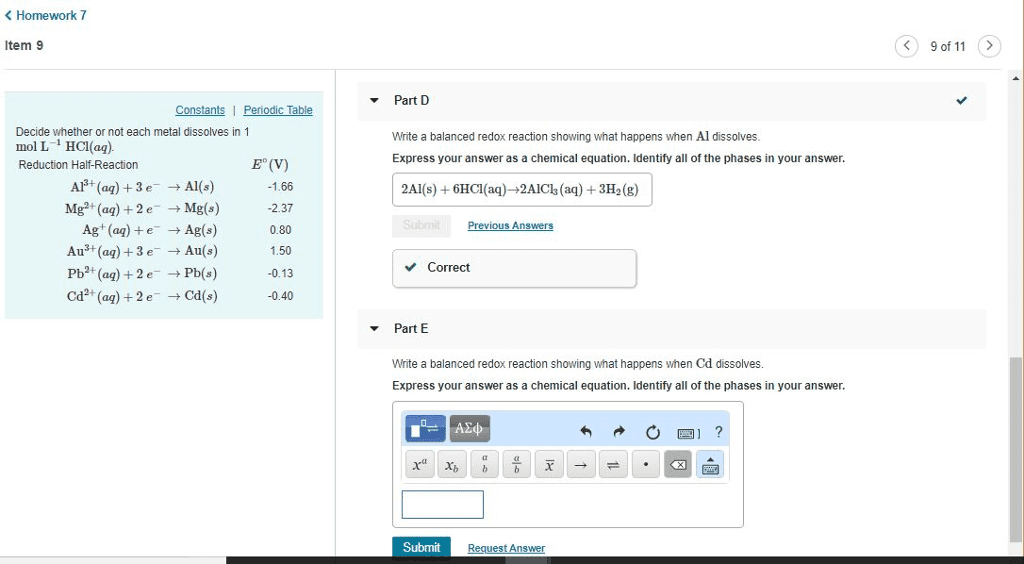
Reduction Half-Reaction Eâ(V) Al3+(aq)+3eâ âAl(s) -1.66 Mg2+(aq)+2eâ âMg(s) -2.37 Ag+(aq)+eâ âAg(s) 0.80 Au3+(aq)+3eâ âAu(s) 1.50 Pb2+(aq)+2eâ âPb(s) -0.13 Cd2+(aq)+2eâ âCd(s) -0.40
Decide whether or not each metal dissolves in 1 molLâ1HCl(aq).
1. Write a balanced redox reaction showing what happens when Al dissolves.
2. Write a balanced redox reaction showing what happens when Cd dissolves.
| Reduction Half-Reaction | Eâ(V) | |
| Al3+(aq)+3eâ | âAl(s) | -1.66 |
| Mg2+(aq)+2eâ | âMg(s) | -2.37 |
| Ag+(aq)+eâ | âAg(s) | 0.80 |
| Au3+(aq)+3eâ | âAu(s) | 1.50 |
| Pb2+(aq)+2eâ | âPb(s) | -0.13 |
| Cd2+(aq)+2eâ | âCd(s) | -0.40 |
Decide whether or not each metal dissolves in 1 molLâ1HCl(aq).
1. Write a balanced redox reaction showing what happens when Al dissolves.
2. Write a balanced redox reaction showing what happens when Cd dissolves.
Casey DurganLv2
24 May 2019