1
answer
0
watching
285
views
11 Nov 2019
Draw Lewis structures for the following:
(a) SiH4 (b) SF2 (c) H2O2 (d) AsO33- (e) H2CO (both H atoms are bonded to C)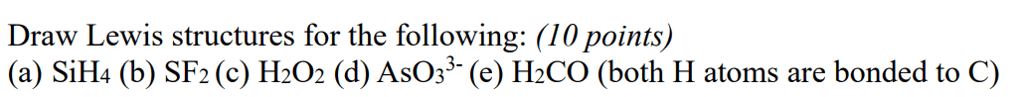
Draw Lewis structures for the following: (10 points) (a) SiH4 (b) SF2 (c) H202 (d) AsOs3(e) H2CO (both H atoms are bonded to C)
Draw Lewis structures for the following:
(a) SiH4 (b) SF2 (c) H2O2 (d) AsO33- (e) H2CO (both H atoms are bonded to C)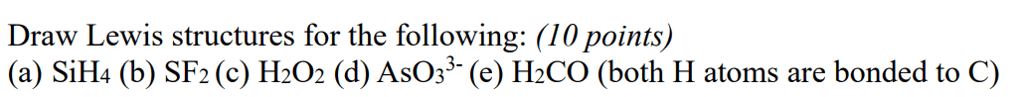
Draw Lewis structures for the following: (10 points) (a) SiH4 (b) SF2 (c) H202 (d) AsOs3(e) H2CO (both H atoms are bonded to C)
Elin HesselLv2
3 Aug 2019
