1
answer
1
watching
810
views
28 Sep 2019
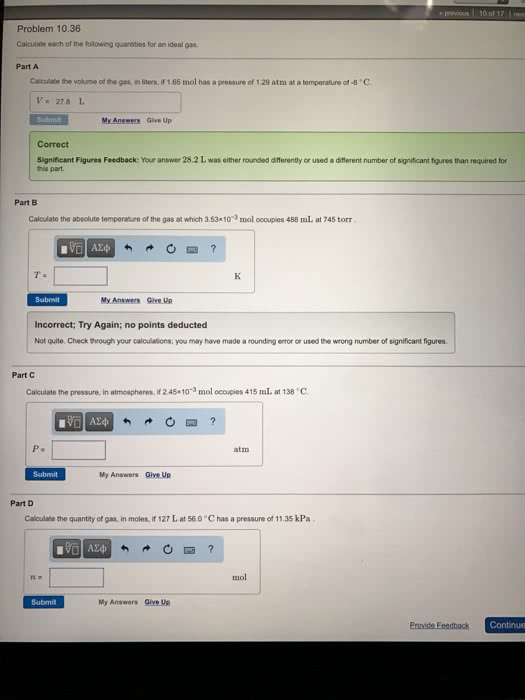
Calculate each of the following quantities for an ideal gas. (a) the volume of the gas, in liters, if 1.73 mol has a pressure of 0.992 atm at a temperature of -6°C (b) the absolute temperature of the gas at which 3.33 10-3 mol occupies 247 mL at 715 torr (c) the pressure, in atmospheres, if 0.0455 mol occupies 428 mL at 125°C (d) the quantity of gas, in moles, if 66.0 L at 57°C has a pressure of 11.27 kPa
Calculate each of the following quantities for an ideal gas. (a) the volume of the gas, in liters, if 1.73 mol has a pressure of 0.992 atm at a temperature of -6°C (b) the absolute temperature of the gas at which 3.33 10-3 mol occupies 247 mL at 715 torr (c) the pressure, in atmospheres, if 0.0455 mol occupies 428 mL at 125°C (d) the quantity of gas, in moles, if 66.0 L at 57°C has a pressure of 11.27 kPa
Jean KeelingLv2
28 Sep 2019