1
answer
0
watching
298
views
11 Nov 2019
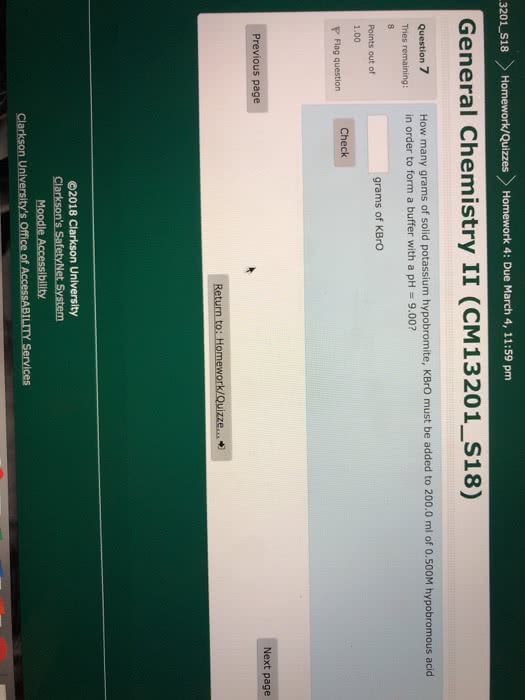

3201 S18 Homework/Quizzes Homework 4: Due March 4, 11:59 pm General Chemistry II (CM13201_S18) How many grams of solid potassium hypobromite, KBrO must be added to 200.0 ml of 0.500M hypobromous acid in order to form a buffer with a pH = 9.00? Question 7 grams of KBro Points out of 1.00 Check Flag question Previous page Next page ©2018 Clarkson University
Nestor RutherfordLv2
14 Jan 2019


