1
answer
0
watching
115
views
11 Nov 2019
The answer is B. Please explain. Thanks!

The answer is B. Please explain. Thanks!


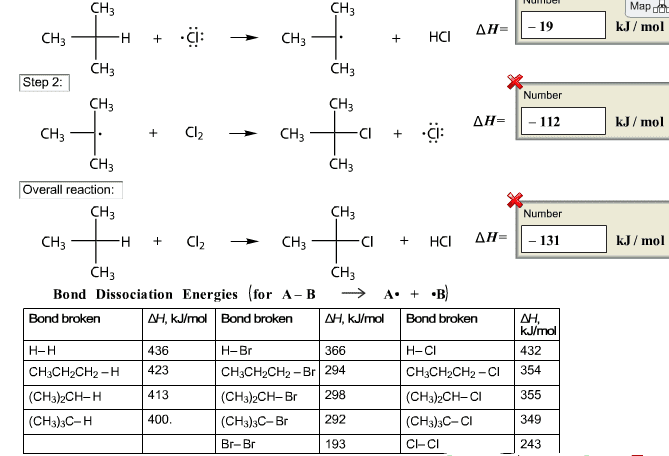
15. Using the information shown below, calculate Î1Pnn for the chlorination of methane to form carbon tetrachloride. CI H-C-H 4 Cl-Cl- + cI Bond Energy (k.J/mol) H-C C1-CI C-Cl H-CI 413 243 339 427 (a) 112 kJ (b) -440 kJ (c) -110 kJ (d) -911 kJ (e) 110 kJ
1
answer
0
watching
115
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Casey DurganLv2
22 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232