1
answer
0
watching
93
views
16 Nov 2019


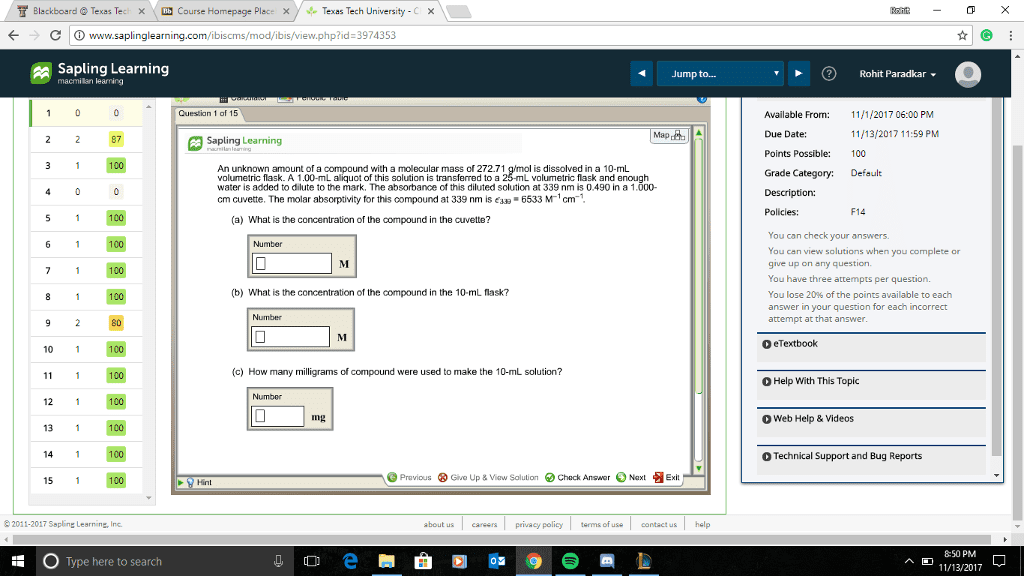
Question 1 (of 10) value: 0.75 points 3 attempts left Check my work Enter your answer in the provided borx Calculate the molar concentration of a sodium hydroxide solution if 49.87 mL of this solution we required to neutralize 17.81 mL of a 0.2000 M solution of hydrochloric acid. â¡M NaOH Type here to search ç¢0å lp
1
answer
0
watching
93
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Tod ThielLv2
21 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232