1
answer
0
watching
215
views
16 Nov 2019
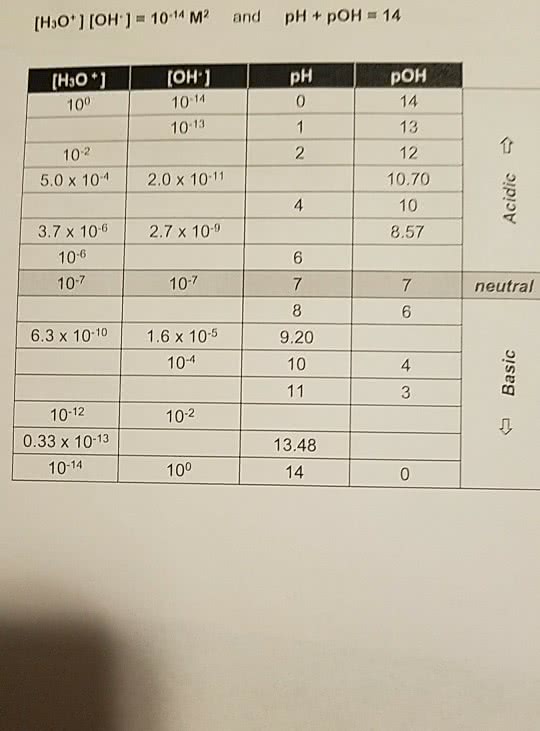
[H30+ ] [OH'] = 10-14 M2 and pH + pOH 14 (HsO 1 100 OH 1 10-14 10-13 pH 0 POH 14 13 12 10.70 10 8.57 10-2 5.0 x 104 2.0 x 10-11 4 3.7 x 106 2.7x 10-9 10-6 10-7 6 10-7 neutral 6 6.3 x 10-10 1.6 x 10-5 9.20 10 10-4 4 3 10-12 0.33 x 10-13 10-14 10-2 13.48 14 100

[H30+ ] [OH'] = 10-14 M2 and pH + pOH 14 (HsO 1 100 OH 1 10-14 10-13 pH 0 POH 14 13 12 10.70 10 8.57 10-2 5.0 x 104 2.0 x 10-11 4 3.7 x 106 2.7x 10-9 10-6 10-7 6 10-7 neutral 6 6.3 x 10-10 1.6 x 10-5 9.20 10 10-4 4 3 10-12 0.33 x 10-13 10-14 10-2 13.48 14 100
1
answer
0
watching
215
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Sixta KovacekLv2
18 Feb 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
What is the molar solubility of silver sulfide? (See the appendix.)
M
What is the concentration of silver ions in a saturated solution of silver sulfide?
M
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Write the chemical equations for the dissociation of each of the following substances and determine their molar solubilities. (Use the lowest possible coefficients. Include states-of-matter under the given conditions in your answer. See the appendix.)
(a) BaSO4
BaSO4(s) ? Ba2+(aq)+SO
| 2- |
| 4 |
(aq)
Correct.
M
(b) Ba(OH)2
Ba(OH)2(s) ? Ba2+(aq)+2OH-(aq)
Correct.
M
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

