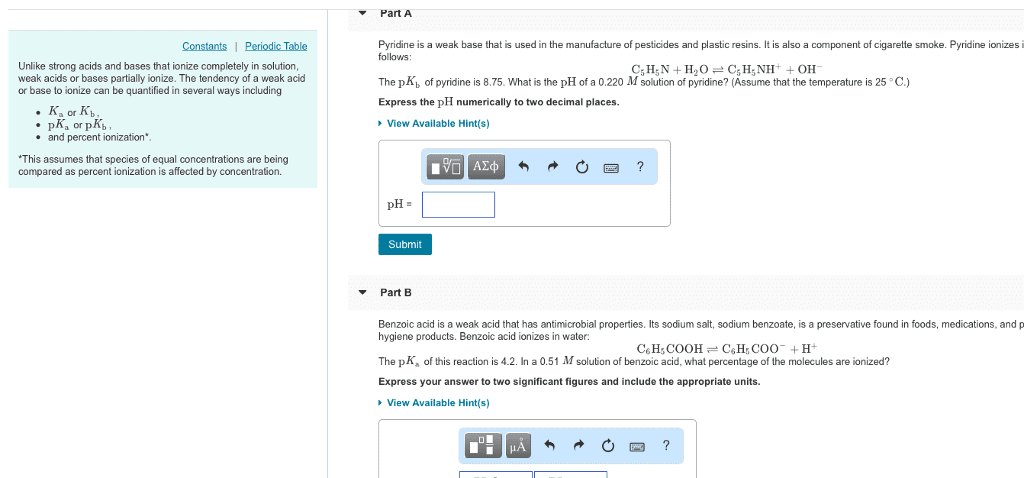
Unlike strong acids and bases that ionize completely in solution, weak acids or bases partially ionize. The tendency of a weak acid or base to ionize can be quantified in several ways including
Ka or Kb ,
pKa or pKb ,
and percent ionization*.
*This assumes that species of equal concentrations are being compared as percent ionization is affected by concentration
Part A
Pyridine is a weak base that is used in the manufacture of pesticides and plastic resins. It is also a component of cigarette smoke. Pyridine ionizes in water as follows:
C5H5N+H2OâC5H5NH++OHâ
The pKb of pyridine is 8.75. What is the pH of a 0.345 M solution of pyridine? (Assume that the temperature is 25 âC .)
Express the pH numerically to two decimal places.
pH=
Part B
Benzoic acid is a weak acid that has antimicrobial properties. Its sodium salt, sodium benzoate, is a preservative found in foods, medications, and personal hygiene products. Benzoic acid ionizes in water:
C6H5COOHâC6H5COOâ+H+
The pKa of this reaction is 4.2. In a 0.77 M solution of benzoic acid, what percentage of the molecules are ionized?
Express your answer to two significant figures and include the appropriate units.
% ionization=
please show steps!
Unlike strong acids and bases that ionize completely in solution, weak acids or bases partially ionize. The tendency of a weak acid or base to ionize can be quantified in several ways including
Ka or Kb ,
pKa or pKb ,
and percent ionization*.
*This assumes that species of equal concentrations are being compared as percent ionization is affected by concentration
Part A
Pyridine is a weak base that is used in the manufacture of pesticides and plastic resins. It is also a component of cigarette smoke. Pyridine ionizes in water as follows:
C5H5N+H2OâC5H5NH++OHâ
The pKb of pyridine is 8.75. What is the pH of a 0.345 M solution of pyridine? (Assume that the temperature is 25 âC .)
Express the pH numerically to two decimal places.
pH=
Part B
Benzoic acid is a weak acid that has antimicrobial properties. Its sodium salt, sodium benzoate, is a preservative found in foods, medications, and personal hygiene products. Benzoic acid ionizes in water:
C6H5COOHâC6H5COOâ+H+
The pKa of this reaction is 4.2. In a 0.77 M solution of benzoic acid, what percentage of the molecules are ionized?
Express your answer to two significant figures and include the appropriate units.
% ionization=
please show steps!