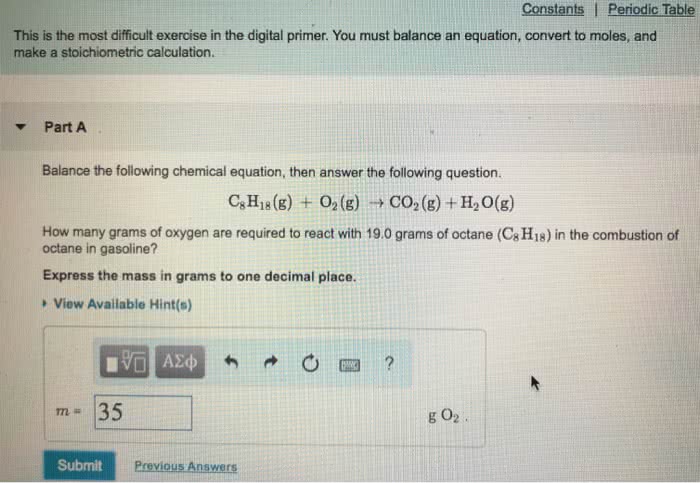
1
answer
0
watching
168
views
18 Nov 2019
Hi, I need help with chemistry... got a little stuck.
Required: Answer the following questions. Make sure to show all work necessary.
a. How many moles are there in 685 grams of N2O?
b. How many moles are there in 592 grams (about 750 mL) of ethanol, C2H6O?
c. One simple way to represent gasoline is as octane, C8H18. A gallon of octane has a mass
of approximately 2.73 kilograms. How many moles are in one gallon of octane,
C8H18? (Hint: First determine how many grams of octane there are in a gallon)
d. How many moles of CO2 are there in a metric ton (1000 kg) of CO2? (Hint: First convert
from metric tons to grams)
Hi, I need help with chemistry... got a little stuck.
Required: Answer the following questions. Make sure to show all work necessary.
a. How many moles are there in 685 grams of N2O?
b. How many moles are there in 592 grams (about 750 mL) of ethanol, C2H6O?
c. One simple way to represent gasoline is as octane, C8H18. A gallon of octane has a mass
of approximately 2.73 kilograms. How many moles are in one gallon of octane,
C8H18? (Hint: First determine how many grams of octane there are in a gallon)
d. How many moles of CO2 are there in a metric ton (1000 kg) of CO2? (Hint: First convert
from metric tons to grams)
1
answer
0
watching
168
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Hubert KochLv2
18 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232