1
answer
0
watching
298
views
19 Nov 2019
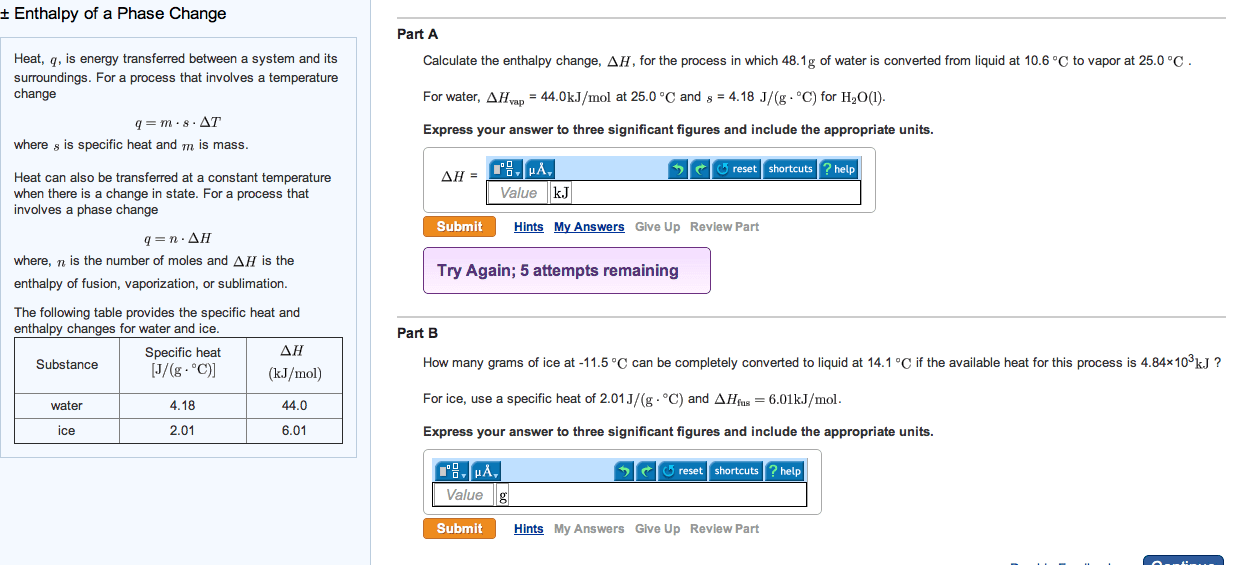
How many grams of ice at -27.8 âC can be completely converted to liquid at 22.2 âC if the available heat for this process is 4.75Ã103 kJ ?
For ice, use a specific heat of 2.01 J/(gâ âC) and ÎHfus=6.01kJ/mol.
How many grams of ice at -27.8 âC can be completely converted to liquid at 22.2 âC if the available heat for this process is 4.75Ã103 kJ ?
For ice, use a specific heat of 2.01 J/(gâ âC) and ÎHfus=6.01kJ/mol.
1
answer
0
watching
298
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
20 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232