1
answer
0
watching
132
views
23 Nov 2019
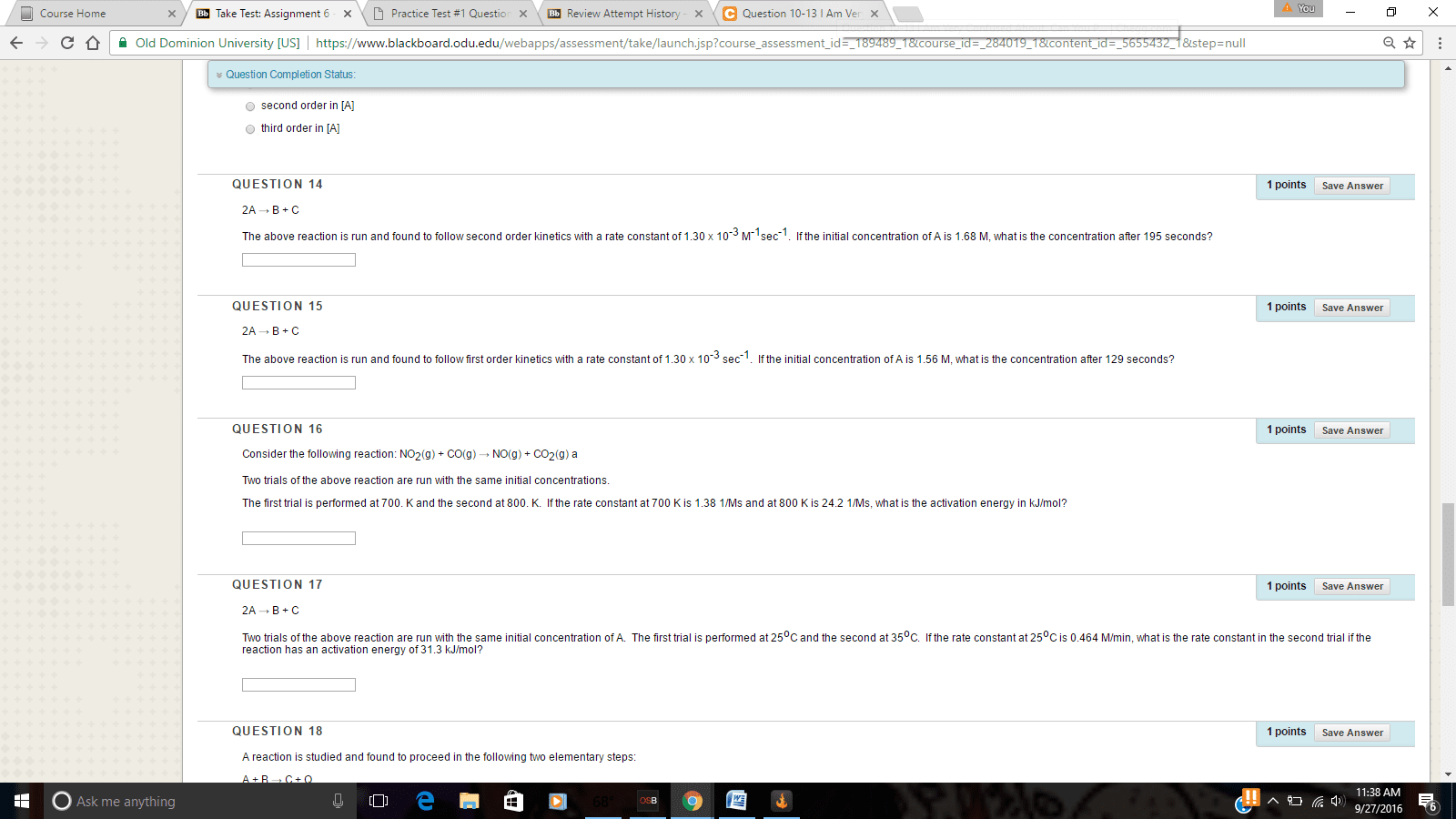
1. 2A â B + C
The above reaction is run and found to follow zero order kinetics with a rate constant of 1.30 x 10-3 Mâ¢sec-1. If the initial concentration of A is 1.34 M, what is the concentration after 132 seconds?
2. 2A â B + C
The above reaction is run and found to follow second order kinetics with a rate constant of 1.30 x 10-3 M-1sec-1. If the initial concentration of A is 1.45 M, what is the concentration after 142 seconds?
1. 2A â B + C
The above reaction is run and found to follow zero order kinetics with a rate constant of 1.30 x 10-3 Mâ¢sec-1. If the initial concentration of A is 1.34 M, what is the concentration after 132 seconds?
2. 2A â B + C
The above reaction is run and found to follow second order kinetics with a rate constant of 1.30 x 10-3 M-1sec-1. If the initial concentration of A is 1.45 M, what is the concentration after 142 seconds?
Jamar FerryLv2
8 Mar 2019