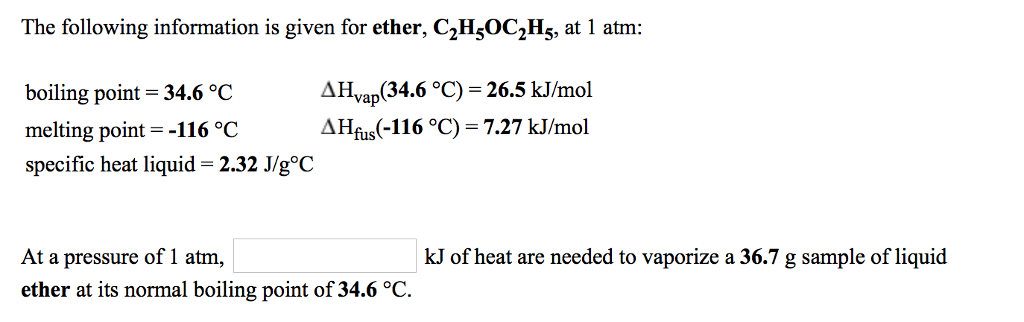1
answer
0
watching
288
views
23 Nov 2019
Use the following information to answer questions 2 through 8:
Diethyl ether, CH3CH2OCH2CH3 ,
specific heat solid:
1.89 J/g K
heat of fusion:
7.19 kJ/mol
normal melting point:
â116.3 °C
Molar mass:
74.1 g/mole
specific heat liquid:
2.32 J/g K
heat of vaporization:
27.2 kJ/mol
normal boiling point:
34.6 °C
specific heat gas:
1.61 J/g K
3. What is the vapor pressure at 25°C?
< 1 atm
= 1 atm
> 1 atm
Use the following information to answer questions 2 through 8:
Diethyl ether, CH3CH2OCH2CH3 ,
| specific heat solid: 1.89 J/g K | heat of fusion: 7.19 kJ/mol | normal melting point: â116.3 °C | Molar mass: 74.1 g/mole |
| specific heat liquid: 2.32 J/g K | heat of vaporization: 27.2 kJ/mol | normal boiling point: 34.6 °C | |
| specific heat gas: 1.61 J/g K |
3. What is the vapor pressure at 25°C?
| < 1 atm |
| = 1 atm |
| > 1 atm |
Nelly StrackeLv2
2 Oct 2019