1
answer
0
watching
174
views
27 Nov 2019
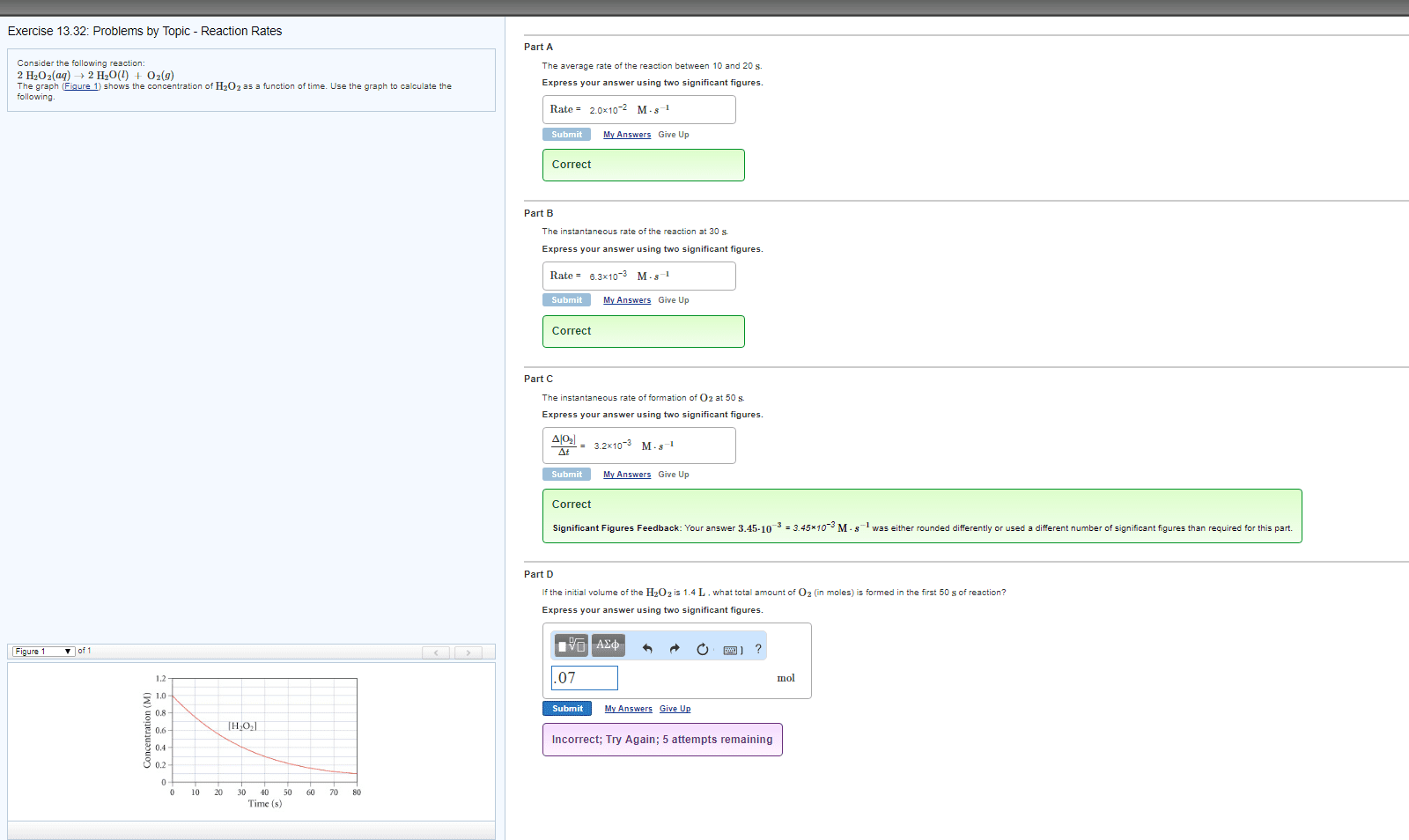
Consider the following reaction:
2H2O2(aq)?2H2O(l)+O2(g)
The graph (Figure1) shows the concentration of H2O2asa function of time. Use the graph to calculate thefollowing.
the initial volume of the H2O2solution is 1.3L, what total amount of O2(in moles) is formed in the first 50 s of reaction? Express your answer using two significant figures.
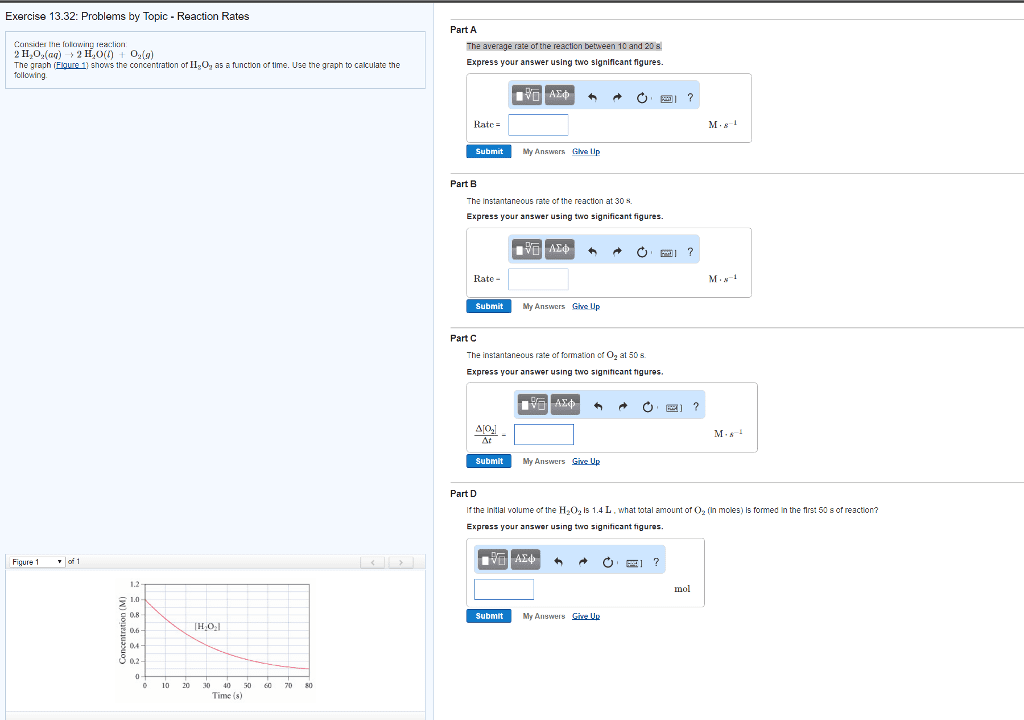
Consider the following reaction:
2H2O2(aq)?2H2O(l)+O2(g)
The graph (Figure1) shows the concentration of H2O2asa function of time. Use the graph to calculate thefollowing.
the initial volume of the H2O2solution is 1.3L, what total amount of O2(in moles) is formed in the first 50 s of reaction?
Express your answer using two significant figures.
Trinidad TremblayLv2
1 Aug 2019