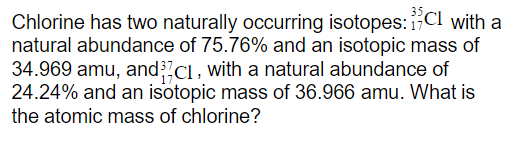1
answer
0
watching
54
views
27 Nov 2019
provide data for Chlorine:
a. M for Cl-37
b. A for Cl-37
c. atomic mass of naturally occuring Cl
suposed answers are:
a. M=37
b. A-36.9659026
c. atomic mass = 35.453
i know how they got a and c, but not b
provide data for Chlorine:
a. M for Cl-37
b. A for Cl-37
c. atomic mass of naturally occuring Cl
suposed answers are:
a. M=37
b. A-36.9659026
c. atomic mass = 35.453
i know how they got a and c, but not b
Hubert KochLv2
23 Mar 2019