0
answers
0
watching
179
views
29 Nov 2019
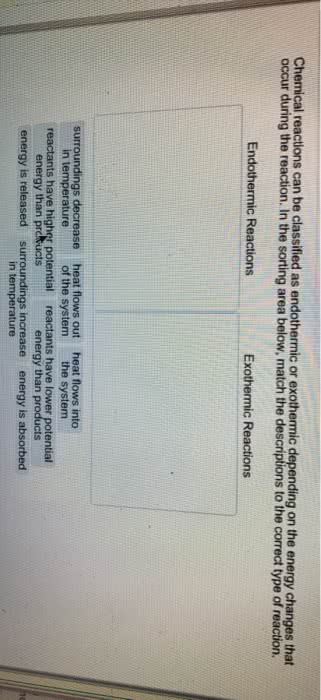
In a gas expansion, 87 J of heat is absorbed from the surroundings and the energy of the system decreases by 128 J. Calculate the work done, in J. Is this reaction exothermic or endothermic?
In a gas expansion, 87 J of heat is absorbed from the surroundings and the energy of the system decreases by 128 J. Calculate the work done, in J. Is this reaction exothermic or endothermic?
0
answers
0
watching
179
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
A system loses 56.8 J of energy during a process. For each work process below determine if the process exothermic, endothermic, or adiabatic?
| it does 125.0 J of work | Answer 1 Choose...endothermic, exothermic, adiabatic |
| it has 25.0 J of work done on it | Answer 2 Choose...endothermic, exothermic, adiabatic |