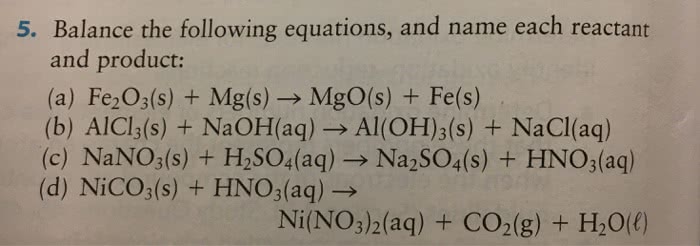1
answer
0
watching
220
views
11 Dec 2019
Express the following equation into word equations and write the corresponding net ionic equation.
(a). Zn(s) + HCl(aq) 
(b). HNO3(aq) + CaCO3(s) 
Balance the following equations.
(c). Fe2O3(s) + Mg(s) 
(d). NaNO3(s) + H2SO4(aq) 
(e). BF3(g) + H2O(l) 
Express the following equation into word equations and write the corresponding net ionic equation.
(a). Zn(s) + HCl(aq) 
(b). HNO3(aq) + CaCO3(s) 
Balance the following equations.
(c). Fe2O3(s) + Mg(s) 
(d). NaNO3(s) + H2SO4(aq) 
(e). BF3(g) + H2O(l) 
Analyn TolentinoLv10
28 Nov 2020