1
answer
0
watching
340
views
11 Dec 2019
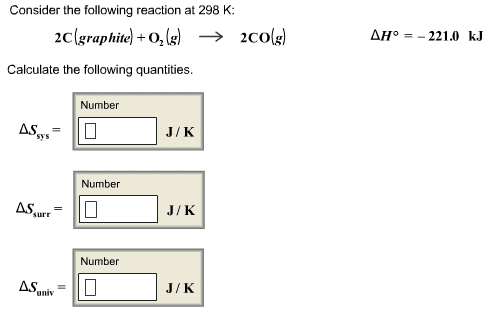
Consider the reaction between graphite and oxygen gas to form carbon monoxide:
2C (s) graphite + O2 (g) → 2CO (g) ΔHrxn = −221.0kJ
- Calculate the entropy change in the surroundings associated with this reaction occurring at 25 ∘C.
- Determine the sign of the entropy change for the system.
- Determine the sign of the entropy change for the universe.
- Is the reaction spontaneous?
Consider the reaction between graphite and oxygen gas to form carbon monoxide:
2C (s) graphite + O2 (g) → 2CO (g) ΔHrxn = −221.0kJ
- Calculate the entropy change in the surroundings associated with this reaction occurring at 25 ∘C.
- Determine the sign of the entropy change for the system.
- Determine the sign of the entropy change for the universe.
- Is the reaction spontaneous?
1
answer
0
watching
340
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Robert KubaraLv10
4 Jan 2021
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232